Molecular representation
We will show you different molecule representations. We will take one molecule and see several ways how we can show it.
This lesson is a part of MEL VR Science Simulations. Learn more →
Similar lessons
Transcript
Have you ever seen a molecule in real life?
Surely not; they are too small.
Most molecules cannot be seen even with the best light microscope. So what do they really look like?

Today in our lab we have aspirin pills.
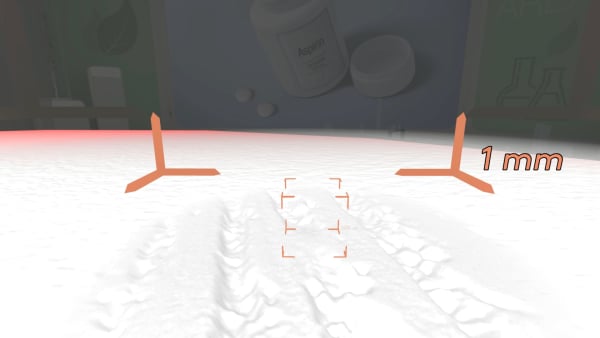
Let's look inside.
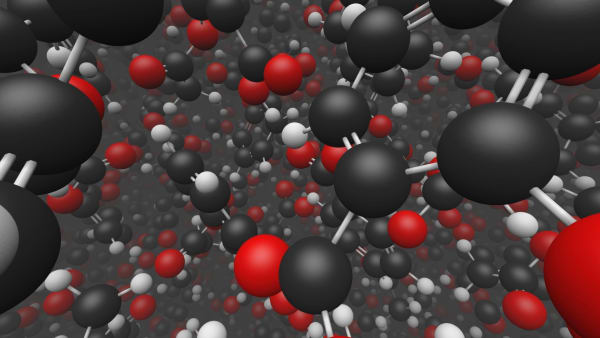
You can see the molecules, that aspirin consists of.
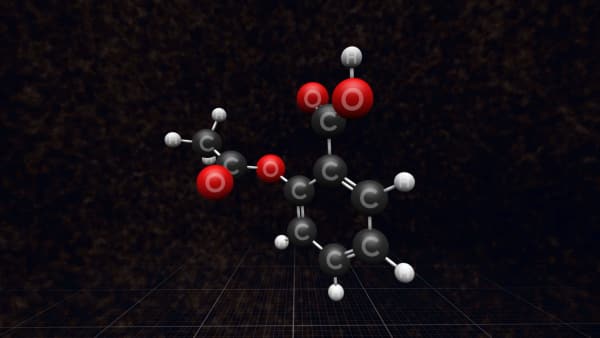
Let's take one of them and look closer. Each ball is an atom and the sticks are the bonds between the atoms that hold them together.
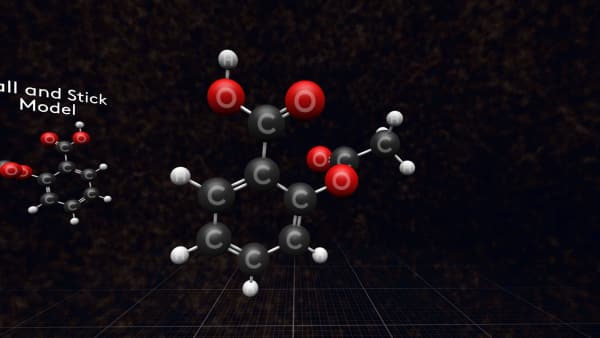
But this is not what a real molecule looks like. It’s just a model that we call a 'ball-and-stick' model.

In fact, the atoms are larger in size and in a molecule, they overlap each other. So, a more accurate model will look like this. We call it a 'space-filling' model.
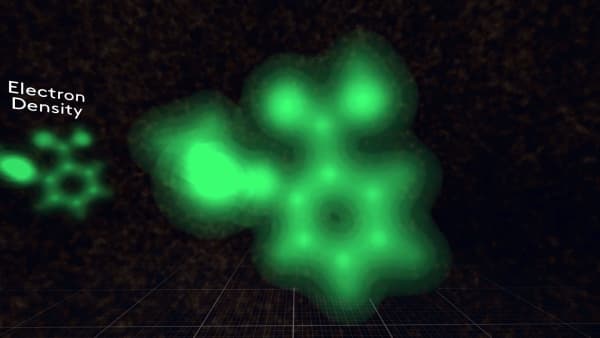
But this is only a model. In a real molecule, electrons are spread among different atoms.
So, if you look at the total electron density in a molecule it will look like this.
Since you cannot see molecules with your eyes, any visual representation is just a model. And different models are more useful for different situations.
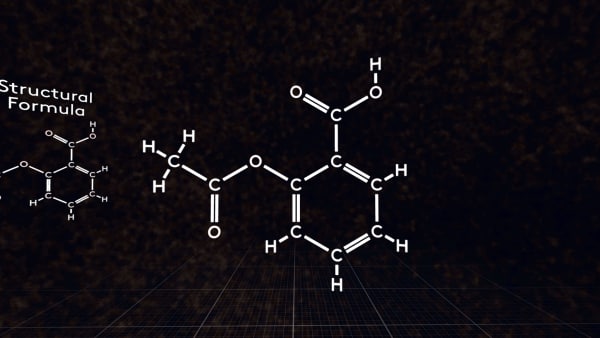
Sometimes it’s more convenient just to use a flat structural formula. For example, when you write the formula on a paper. Like this.
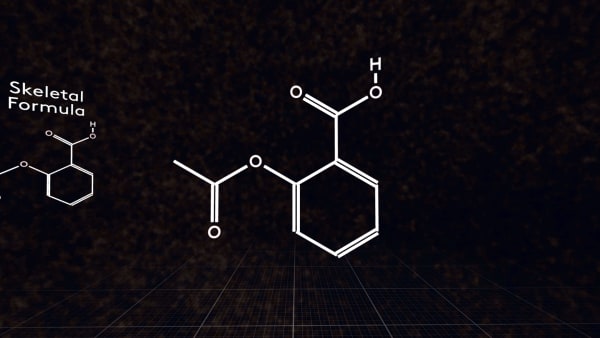
Also for organic compounds, you can write a skeletal formula. For an aspirin molecule, it looks like this.
The skeletal formula is a structural formula in which the hydrogen atoms attached to carbon atoms and carbon atoms themselves are not indicated.
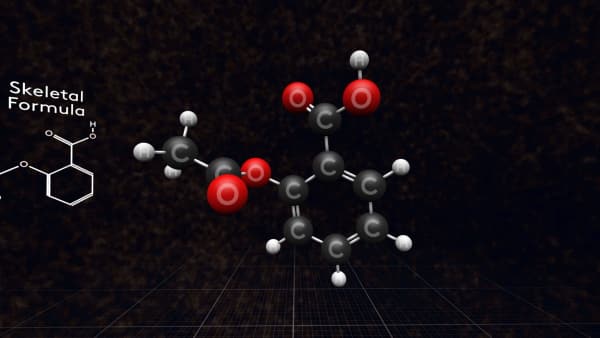
Also, we can just write a molecular formula. Let's see how to make it.
We simply count the numbers of each type of atom in a molecule. So, the aspirin molecule consists of:
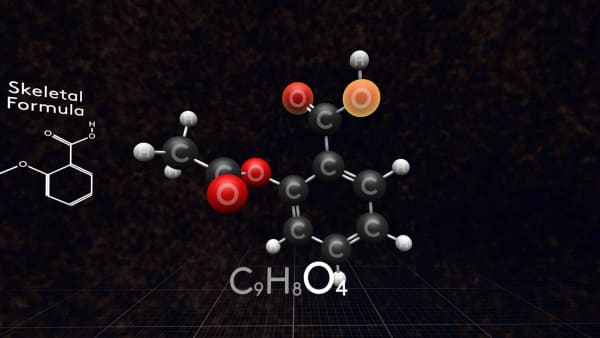
Nine carbon atoms, Eight hydrogen atoms, Four oxygen atoms.
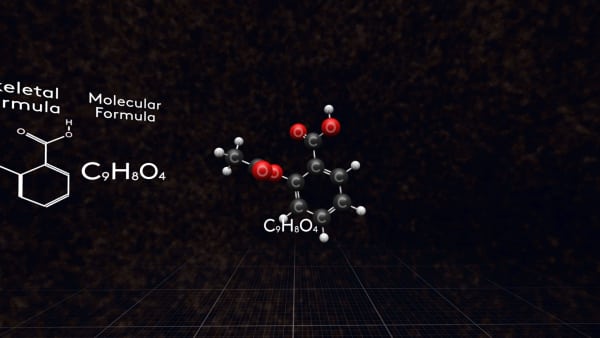
This is the molecular formula of aspirin.
But we have a little problem with molecular formula. Just look!
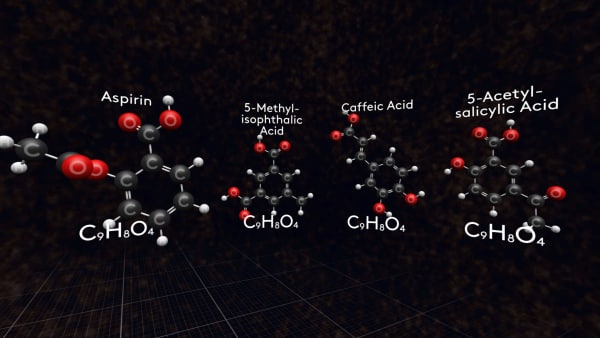
There is another molecule with formula C9H8O4: and another! and another! there are more than 700 molecules with this molecular formula. So, be careful using the molecular formula when talking about a specific substance.
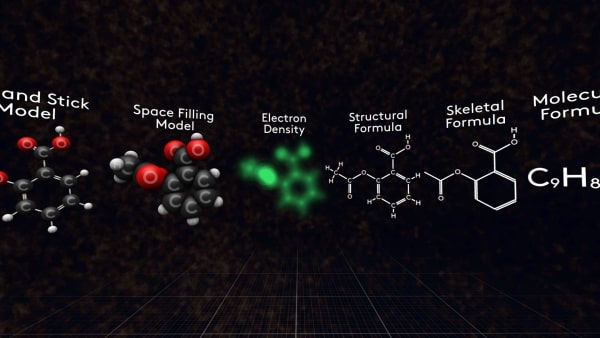
Look at these molecules, these are different representations of aspirin molecules.
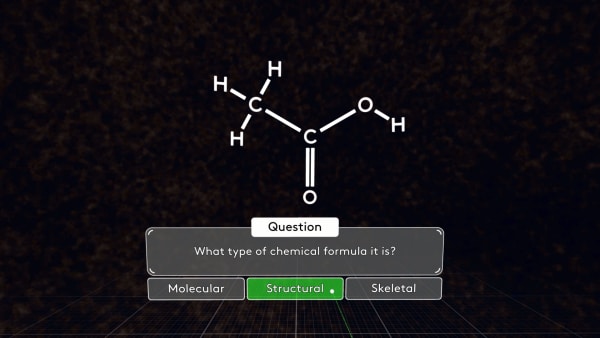
What type of chemical formula is it? Correct! The bonds between particular atoms are shown here, that's why it's a structural formula.
Teacher's notes
Keywords
molecules, atoms, bonds, ball and stick model, space-filling model, electron density model, structural model, skeletal formula, molecular formula
Students will
- Learn different ways to represent molecules
- Find out which representation is suitable for each purpose
Hands-on activities
After VR
Ask students to build ball and stick molecule models.
Topics to discuss
- Tube or rail map analogy: representing real-life objects a certain way, taking into account what information we want to deliver. Compare two maps: a tube map and a city map with tube stations.
- Different ways of representation. How to choose the most suitable representation. Information we can obtain from a particular representation.
- Skeletal formula principle, explanation and examples.
- Why sometimes we need 3D models.
Questions
- Which model to choose if you want to see how many bonds are between atoms in a molecule?
- Why a molecular formula is not a convenient way to describe a molecule?
Calculating
Count how many atoms of each type are in different molecules (from structural models).



