Fox tail
Synthesize nitrogen dioxide and study its properties!


Reagents
Safety
-
Put protective eyewear on.
-
Conduct the experiment on the tray.
-
Make sure Petri dish is clean and dry.
-
Conduct the experiment only in a well-ventilated room.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
I don’t have a clean Petri dish with a blue circle. What to do?
If you have already used the Petri dish with a blue circle in the chlorine Cl2 synthesis experiment, simply rinse it and dry with a paper towel. The other Petri dish would not work for this experiment because the height of the cap placed in the center would not allow you to seal the Petri dish properly.
What’s the template for?
Pick a template appropriate for this experiment and place a Petri dish onto it. Tags by the circles will tell you which chemical has been used at the beginning of the experiment, so that after the experiment you could easily compare the color of compounds in a vial and in a drop.
After waiting for the time specified, I still don’t see the drops change their color. Why?
Make sure you use a Petri dish marked with a blue circle, and that the gas is not leaking through any openings. Also, remember to thoroughly moisten the cap contents with sodium hydrosulfate NaHSO4 solution.
My thymol blue solution is yellow. Is it normal?
It’s okay. A solution of thymol blue may turn yellow as it absorbs carbon dioxide CO2 from air in the vial. You may use this thymol blue solution for your next experiments. However, color of the liquid will change from yellow to red, instead of usual blue to yellow.
Yet, if you would like to obtain a blue solution, pour out all the thymol blue into a plastic cup and add there 1 drop of sodium hydroxide NaOH using a pipette from the previous experimental sets. The solution will turn blue, and you’ll be able to use it in the next experiments.
Step-by-step instructions
-
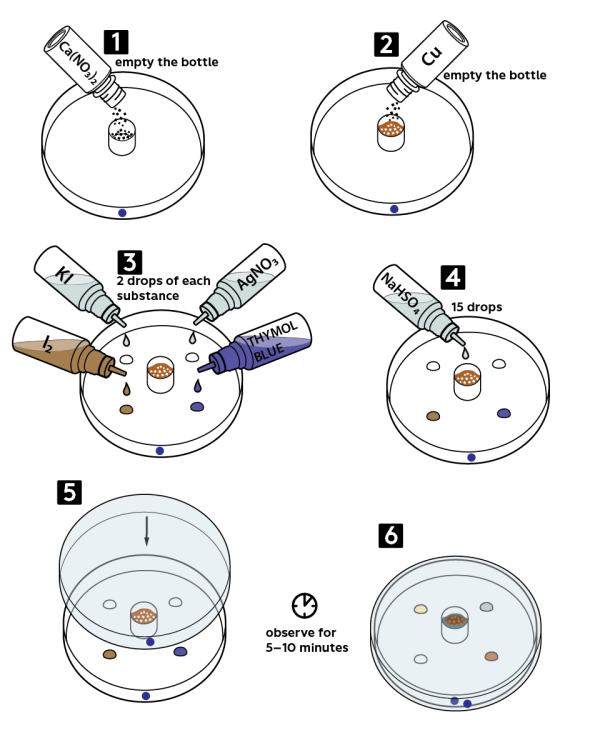
Take a Petri dish with a blue circle on it. Place a white cap from the set onto the center of the Petri dish. Pour all the copper nitrate Cu(NO3)2*4H2O from a vial into the cap.
-
Add there all the copper Cu powder from the vial.
-
Add 2 drops of each reagent: 0.1M potassium iodide KI solution, 0.5% iodine I2 solution, and 0.0002M thymol blue solution.
-
Add 15 drops of 3M sodium hydrosulfate NaHSO4 solution into the cap.
-
Close the Petri dish with a cover marked with a blue circle. Wait 5−10 minutes. In a central drop, bubbles of nitrogen dioxide NO2 will appear.
-
In about 10 minutes, look at the drops of the solutions applied: iodine will lose its color, thymol blue will become yellowish-brown, and potassium iodide will turn yellow.
Disposal
Dispose of the experiment residues along with regular household trash. Dilute the contents of the plastic cap with water before disposing.