Chemical nebulae
Capture a vivid glow in a vial!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
You can purchase hydrogen peroxide at any local pharmacy. Any concentration from 3% to 10% should be fine.
In general, the more luminol you add, the brighter the solution will glow. On the other hand, the glow will fade faster.
Phenol red is a pH indicator and changes colors depending on the acidity of its medium. It turns yellow in an acidic medium ( pH < 6.8), and red in an alkaline medium (pH > 8.2). The phenol red solution in the set is aqueous, and thus is initially orange. But when you add it to the sodium carbonate Na2CO3 solution, it turns red.
Don’t worry, this isn’t critical. Just continue the experiment.
The dark atmosphere makes the result of the experiment much more spectacular. Bright conditions will make the glow more difficult to see.
If you’re performing this experiment in a very dark room and can’t see what you’re doing, turn on a weak light source (dim light or phone light), add the reagent, and turn off the light immediately.
If you followed the instructions carefully, this is likely because your room isn’t dark enough. You need almost complete darkness for the best result. For example, you can conduct the final part of this experiment in a bathroom with all the lights off. Be careful in the darkness and don’t rush!
This actually depends heavily on external conditions like temperature and reagent concentrations. In our case, the luminol glows best for about 30 seconds, and then gradually fades away.
You might notice that even when the glow dies down, other reactions continue taking place in the vial. Some bubbles appear, and the solution gradually pales.
In the presence of iron ions, hydrogen peroxide actually decomposes to yield oxygen gas, which forms bubbles in the solution. The solution’s color changes slightly due to some side processes, primarily the decomposition of the reagents we added.
Step-by-step instructions
First, you'll need a solution of a reducer, luminol, and an oxidizer, hydrogen peroxide H2O2. To create just the right medium for the reaction you're going to observe, add some sodium carbonate Na2CO3. This will make the solution slightly basic.
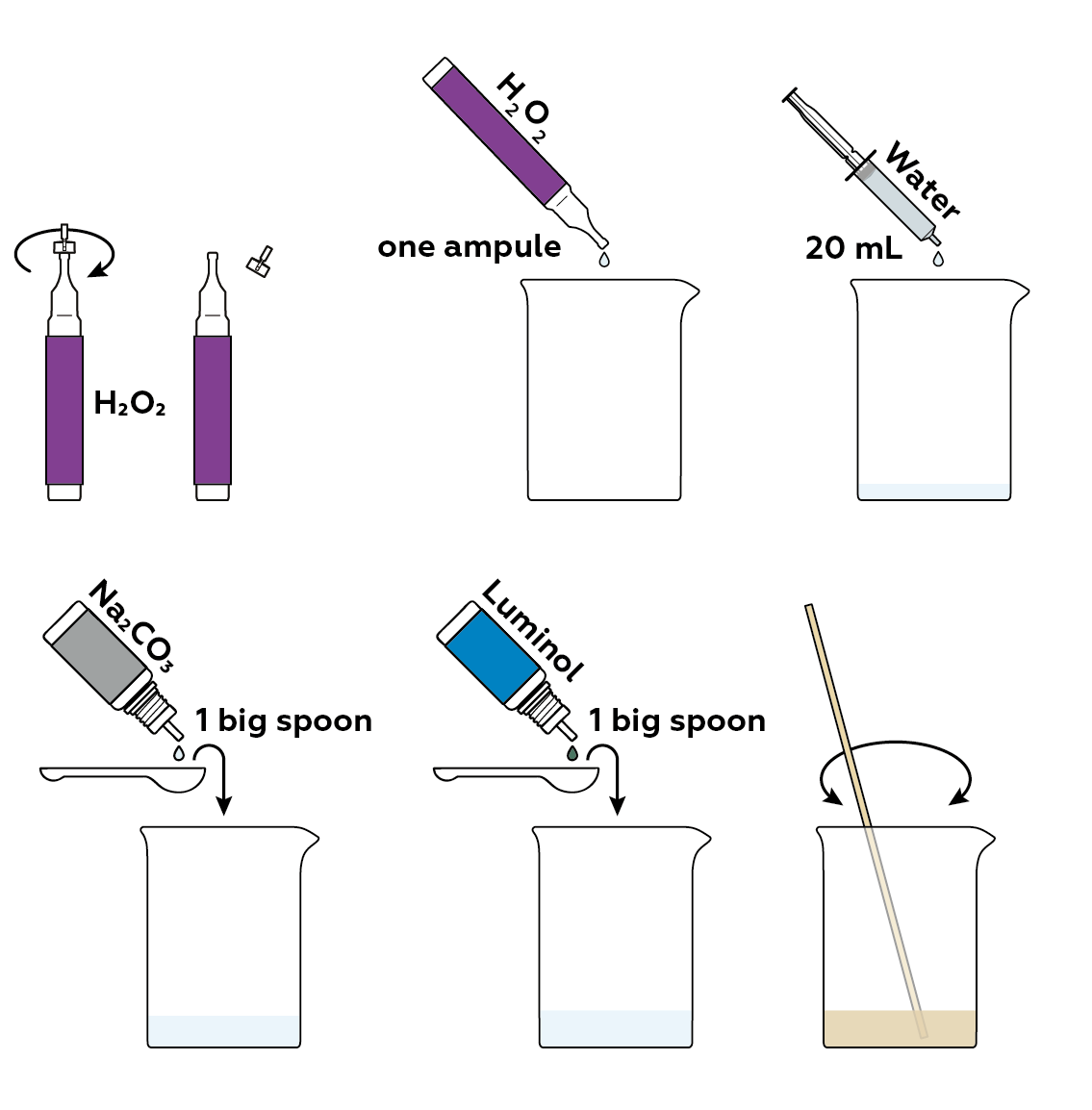
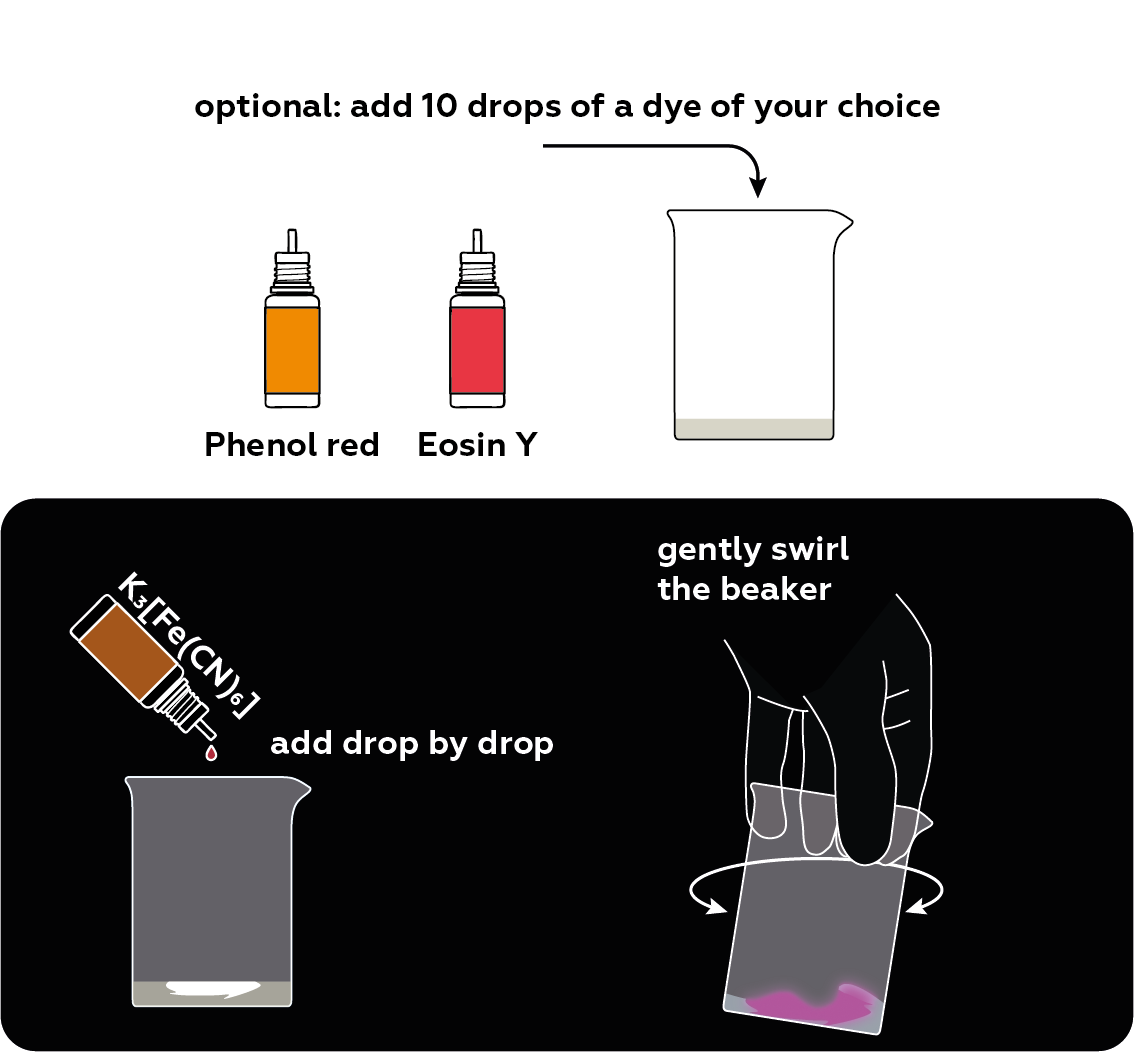
Pour your luminol solution into three vials. Add some dyes.
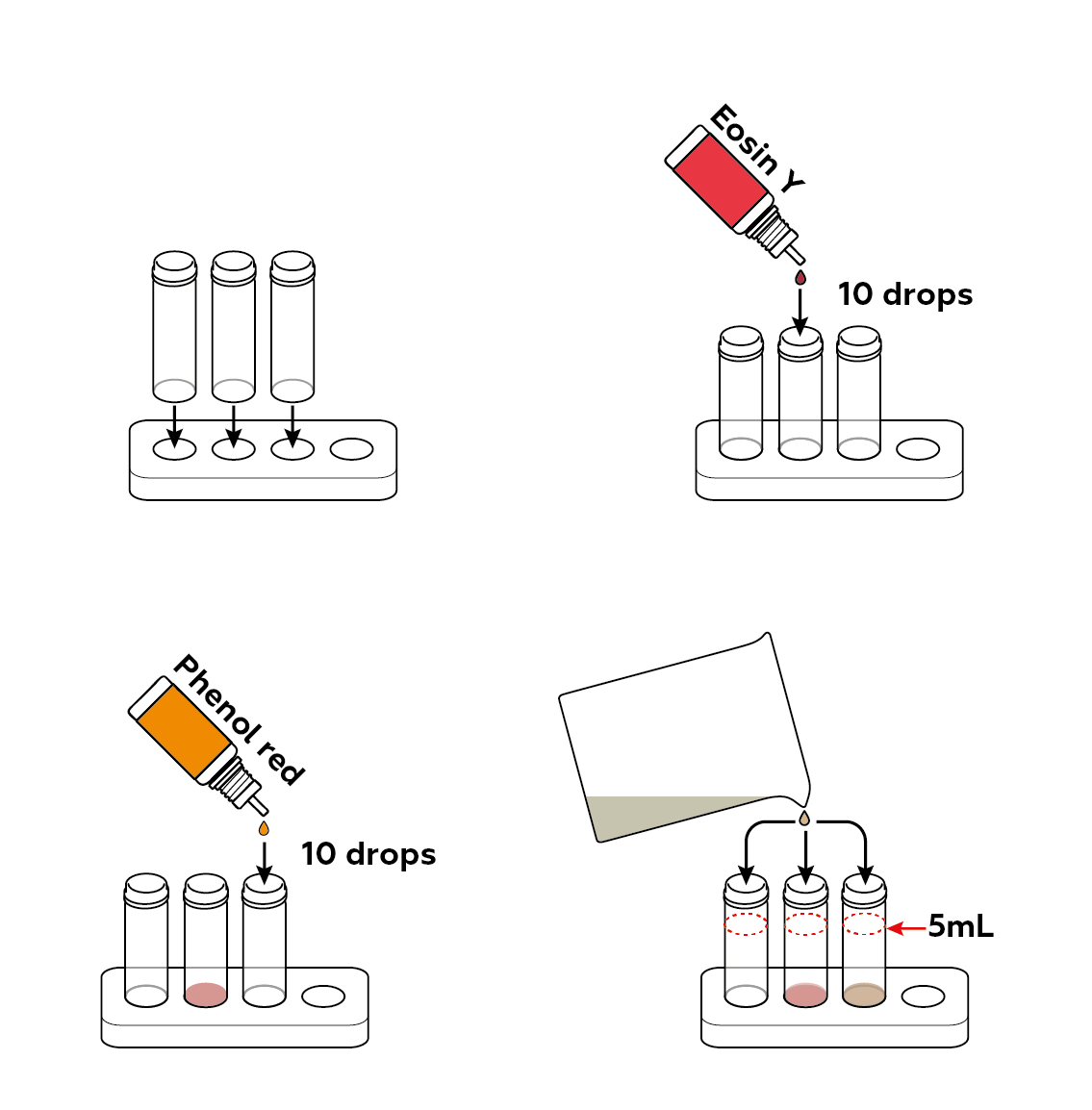
Add some potassium hexacyanoferrate(III) K3[Fe(CN)6]. Observe what happens.

Wash the vials to repeat the experiment. Try and perform the same reaction in your beaker.
Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage. Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description

are made up of various combinations of atoms, which in turn are comprised of nuclei
and “clouds” of electrons
. Some molecules readily give their electrons up, while others would rather take new electrons from other molecules. When one molecule
gives electrons to another
, this is called an oxidation-reduction reaction. The “receiving” molecule is called an oxidizer, while the “giving” molecule is called a reducer.

oxidizes luminol
, taking one of its electrons, the luminol begins to glow. But this reaction needs more than just these two main components; it needs something to facilitate their reaction. In our experiment, potassium hexacyanoferrate(III)
helps H2O2
take electrons away from luminol
, but many compounds of copper Cu, manganese Mn, and some other metals can play this role as well.
Glowing luminol releases blue-violet light, but some dyes, like eosin or phenol red, can absorb blue light and release light of different colors.
That’s interesting!
What makes clock hands visible in the dark?
Watch hands owe their glow to the presence of a phosphor, a substance capable of emitting its excess energy in the form of visible light or ultraviolet rays. When exposed to light, a phosphor’s electrons “jump” temporarily to higher energy levels. As they return to their initial state, they release the excess energy in the form of quanta of light. Our eyes don't see the individual flashes, however – we perceive a continuous stream of light.



