Ion-exchange resin
Make a filter that can purify water of heavy metals!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the safety underlay.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
What should I do?
This isn’t a big deal. To remove the excess resin, pinch the filter at “2.5” mL mark and empty the extra back into the vial. Continue the experiment.
The cylinder shouldn’t stick out the top. Gently push it further in. The resin must be tightly compressed.
You will use it to compare the properties of the unfiltered and filtered solutions. This way, you can check how effectively your filter works!
Yes! You removed the copper using ammonium carbonate, so the resin can be reused. Pour some more copper sulfate solution through the filter and collect the filtrate in a new vial.
This usually happens when the resin isn’t packed in the pipette densely enough. Take another pipette from the set, fill it with resin, and try to compress the resin more firmly using cotton cylinders and a wooden stick.
Step-by-step instructions
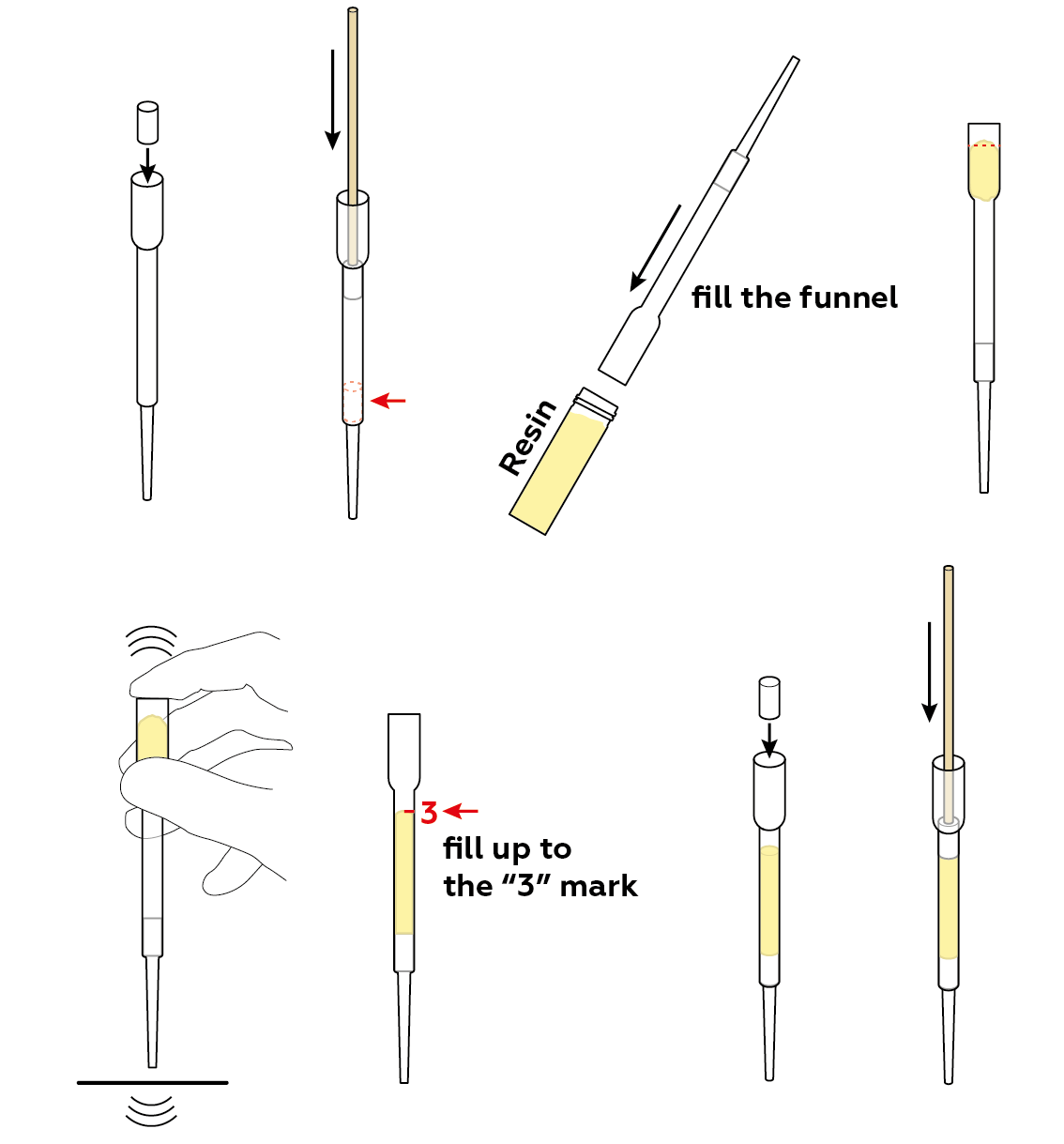
Prepare a filter made of ion-exchange resin. To increase its effectiveness, compress the resin slightly using cotton cylinders and a wooden stick.
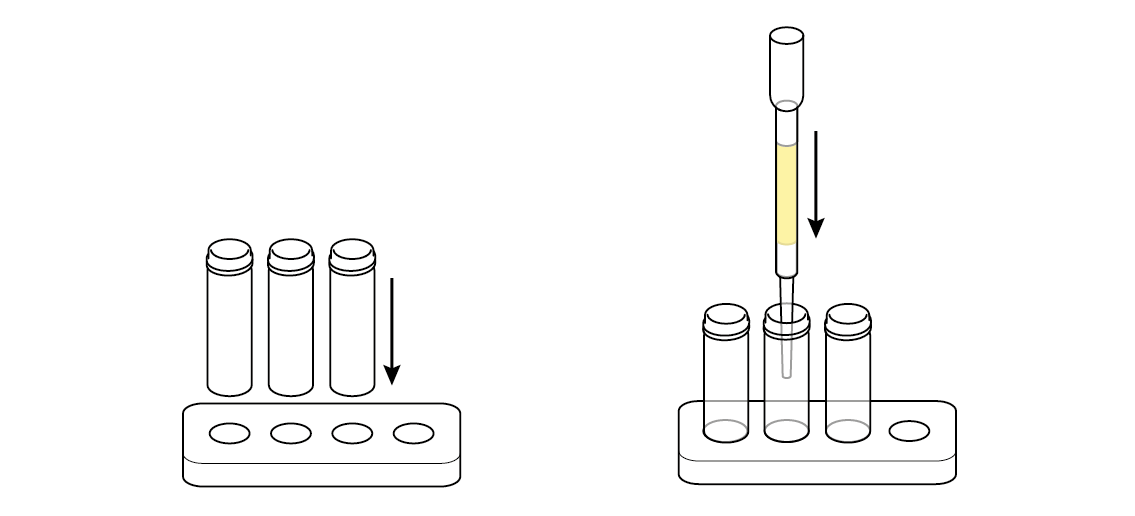
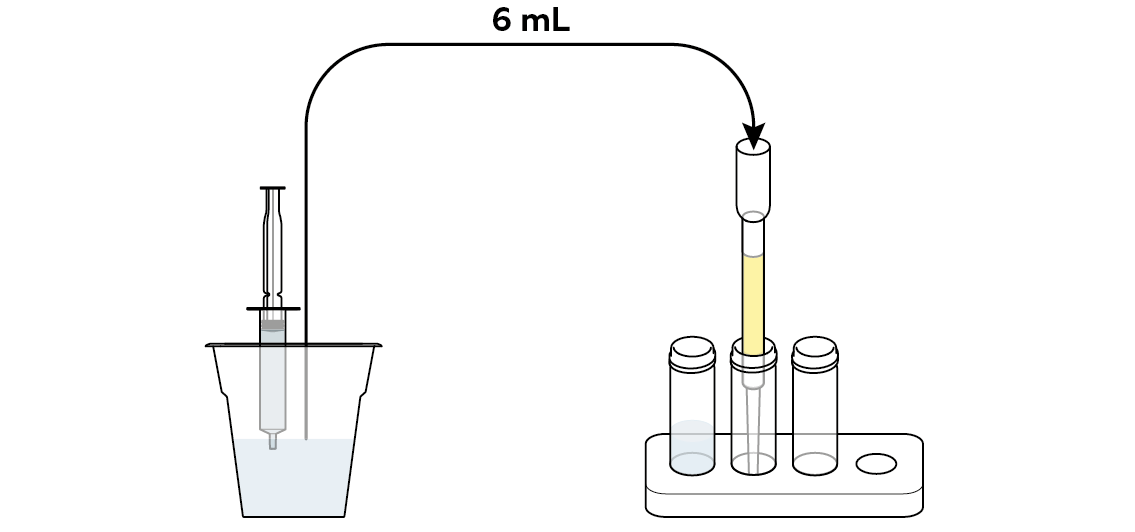
Set up the test stand.
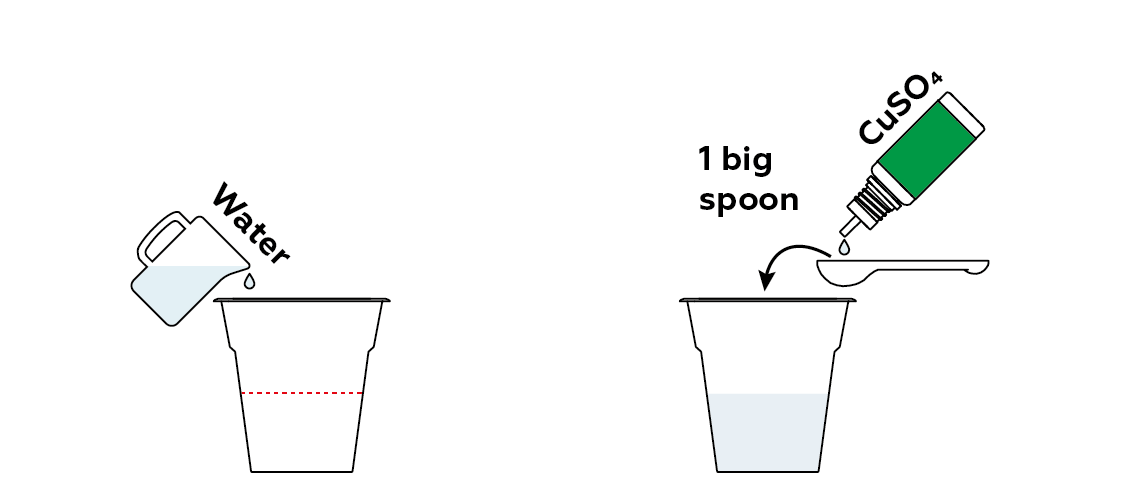
Contaminate some water using copper sulfate CuSO₄. Here, copper acts as our heavy metal.
Pour some of the tainted water straight into the first vial. You will need it later, to help assess the effectiveness of your filter.
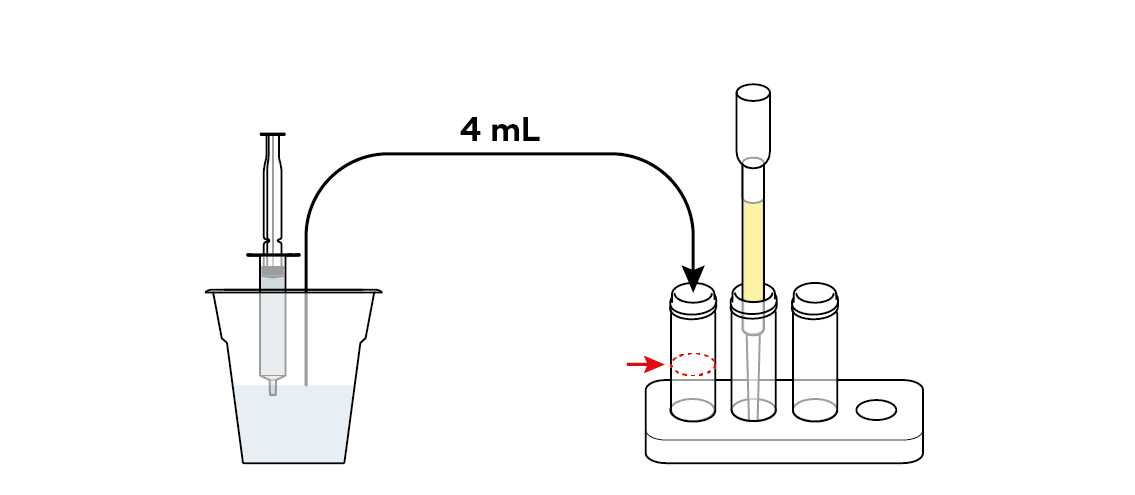
Pour some of the tainted water through the filter into the second vial.
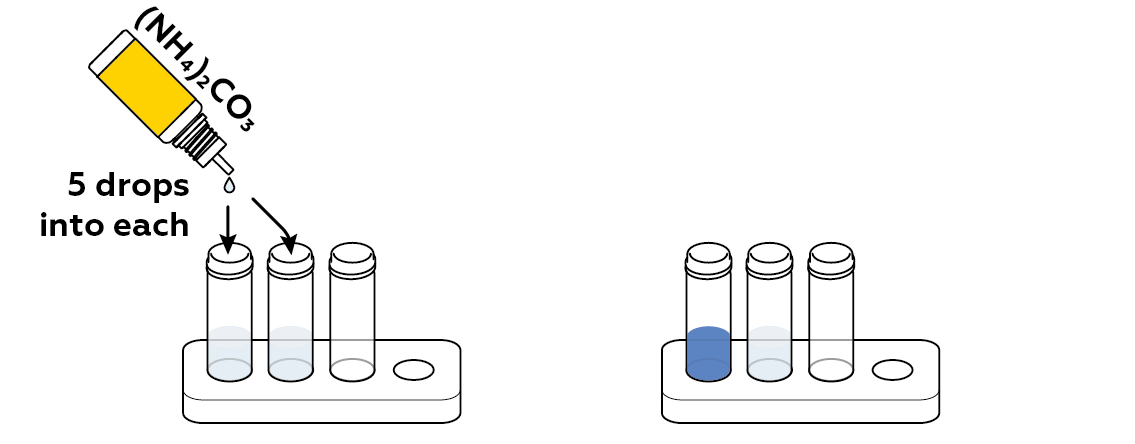
Now, perform a qualitative test for copper ions Cu²+ by adding ammonium carbonate (NH₄)₂CO₃ to the test vials. If a solution contains copper ions, the liquid will turn blue. If your filter succeeded in cleaning the water in the second vial, this water will remain clear.
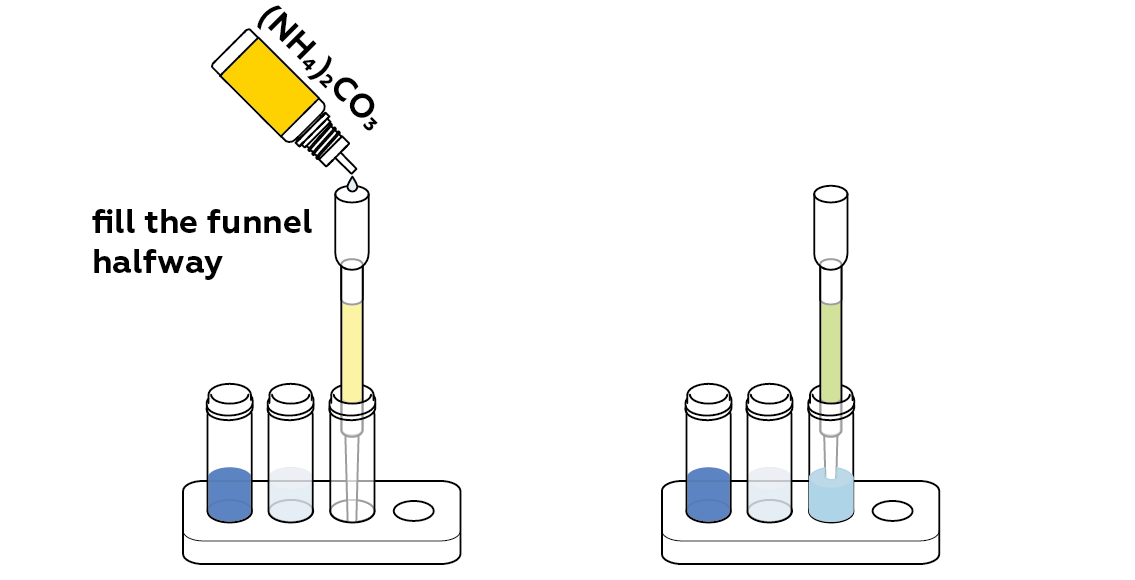
What happens if you apply some ammonium carbonate to the filter?
Wash both vials to repeat the experiment.
Expected result
Your self-made filter with ion-exchange resin purifies water from ions of a heavy metal - copper.
The reaction between ammonium carbonate (NH4)2CO3 and copper ions Cu2+ changes the color of the initial solution from light-blue to deep indigo. The water sample purified through the filter remains colorless, proving the effectiveness of the filter.
Any copper ions left on the ion-exchange resin can be detected by adding a few drops of ammonium carbonate into the filter.
Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage. Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description


We use copper ions 

These resins are long polymers with ions of alkali metals, such as sodium ions 




Though this filter is effective against heavy metals, it is powerless against many organic pollutants 
That’s interesting!
Why do metals replace each other?
The resin works via ion exchange, a reversible chemical process in which ions are exchanged between a solid and a solution. In our case, negatively-charged ions are rigidly attached to the matrix of resin grains. These ions are concentrated on the surface of the resin. They are balanced by positively-charged ions such as sodium Na+, which move freely through the pores of the resin.
In water, copper sulfate CuSO4 splits into ions: positively-charged Cu+2 and negatively-charged SO-4. As the polluted solution passes through the resin, the resin’s surface picks up metal Cu+2 ions, releasing sodium ions Na+ into the solution. Each grain of resin accumulates these charges to a certain limit, after which it must be cleansed of the unnecessary charged particles. To do this, a table salt NaCl solution is passed through the filter to remove the ions from the surface of the resin. The offending ions are washed down the drain, and the free space is repopulated by sodium ions once again. This is called regeneration or recycling of resin.
Heavy metals in water
Heavy metals are a family of chemical elements mostly toxic to humans, especially if they are present in fairly large amounts. In water, they normally exist in the form of positively-charged particles (cations) — take, for example, copper Cu2+, lead Pb2+ or iron Fe3+.
Interestingly, our bodies need certain heavy metals in small quantities, such as iron Fe, zinc Zn, copper Cu, and molybdenum Mo. However, these can be harmful if accumulated in greater quantities. Meanwhile, such heavy metals as lead, cadmium, and mercury are harmful to our bodies even in small amounts.
Many heavy metals are found in water in the form of cations. These must be filtered out for water to be drinkable. We can use an ion-exchange resin filter to accomplish this.