Instant snow
Just add water!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
If you dry your obtained artificial snow thoroughly, it can be used again. However, the sodium polyacrylate will not shrink back to its original compact shape. Therefore, moistening it a second time will not create the same extensive expansion effect as the first time. The set includes several bottles of sodium polyacrylate so you can repeat the experiment with the same dramatic results several times.
Yes, you can, but make sure to wear protective gloves. Never, ever, eat this artificial snow!
Artificial snow can be disposed of just like ordinary waste. If you have dropped or spilled it, you can use a vacuum cleaner or cloth to clean it up. After adding citric acid, drain the resulting solution into the sink and wash with an excess of water.
Step-by-step instructions
The "snow" you're going to make requires just two ingredients: water and a little bit of sodium polyacrylate.
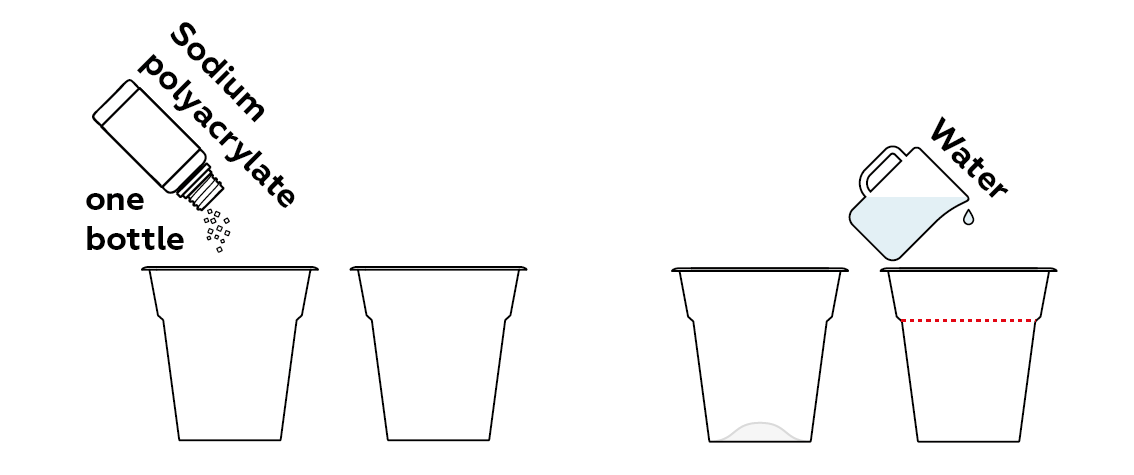
The sodium polyacrylate rapidly absorbs all the water, turning into a heap of fluffy white "snow."

-COO- groups can easily be converted to -COOH groups. You just need to add an acid. These -COOH groups are much worse at binding to water, and the "snow" starts to "melt."
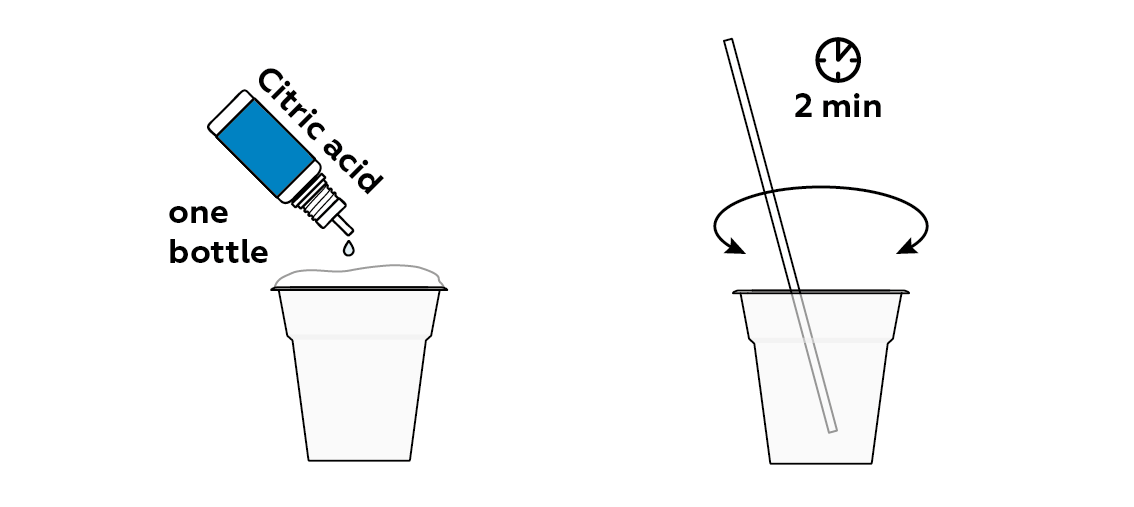
Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage.
Scientific description




—and extremely long polyacrylate chains
. Each chain contains an enormous number of -COO\-
groups.


, but other water molecules make stronger connections to the ones bound to -COO\-
groups. This results in a whole bunch of water molecules attaching firmly to a single -COO\-
group. A single polyacrylate chain can hold 200–300 times its weight in water, which means that a few grams of dry sodium polyacrylate can capture a whole cup of water easily.
What will happen if we add less/more water?
Adding less water will still cause the granules to swell; the experiment will work, but the overall effect won’t be as noticeable. However, be careful not to add too much water: with an excess of Н2O, the granules will stick together and gel, forming a semitransparent, viscous mass similar to jelly.
Follow up
To use the hydrogel beads, put them in a container and add clean water – preferably boiled (but not hot!) or bottled. Tap water can stunt the beads’ "growth."
Place 10–15 hydrogel beads in a disposable cup from the Starter Kit and add a whole cup of water. Let them swell for 6–12 hours. Try not to disturb them during this time — you might damage their structure, which could cause them to fall apart when fully swollen. Fully-grown beads should be spherical. Then, just drain the excess water to start studying and playing with your hydrogel beads. Be sure to wash your hands after playing with them!
Full-size beads can maintain their shape for several days, but will quickly return to their original state if stored outdoors. If this occurs, you can regrow them by reimmersing them in water! If you add some food coloring to the water, you can even tint your beads different colors!
Such hydrogel beads are often used for decoration. But they are also utilized to grow and preserve flowers! A vase with such a filler instead of pure water looks impressive, and the flowers stay healthy and vibrant much longer.
That’s interesting!
What is sodium polyacrylate used for?
Sodium polyacrylate is a superabsorbent polymer. Its colloquial name, "waterlock," is derived from its ability to absorb hundreds of times its mass in water. It has a wide range of applications across different industries due to its ability to absorb huge amounts of water: it is used in medical bandages, diapers, pet pads, and surgery sponges. Moreover, it is utilized as concrete protection and liquid waste control in manufacturing, protection for wires and cables from moisture, and so on. It is also added to containers of different kinds of fuel, such as jet fuel and diesel.
Often, sodium polyacrylate is used to grow or preserve flowers and plants because it lessens the need to water them frequently. You can even try this lifehack at home! In addition, sodium polyacrylate works as a sequestering agent in many cleaning products, as it can bind with substances dissolved in water. This allows cleaning products to act effectively. And of course, as you already know, it is often used as artificial snow!