Chemical frost
A little bit of icy magic!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray.
- Observe safety precautions when working with boiling water.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
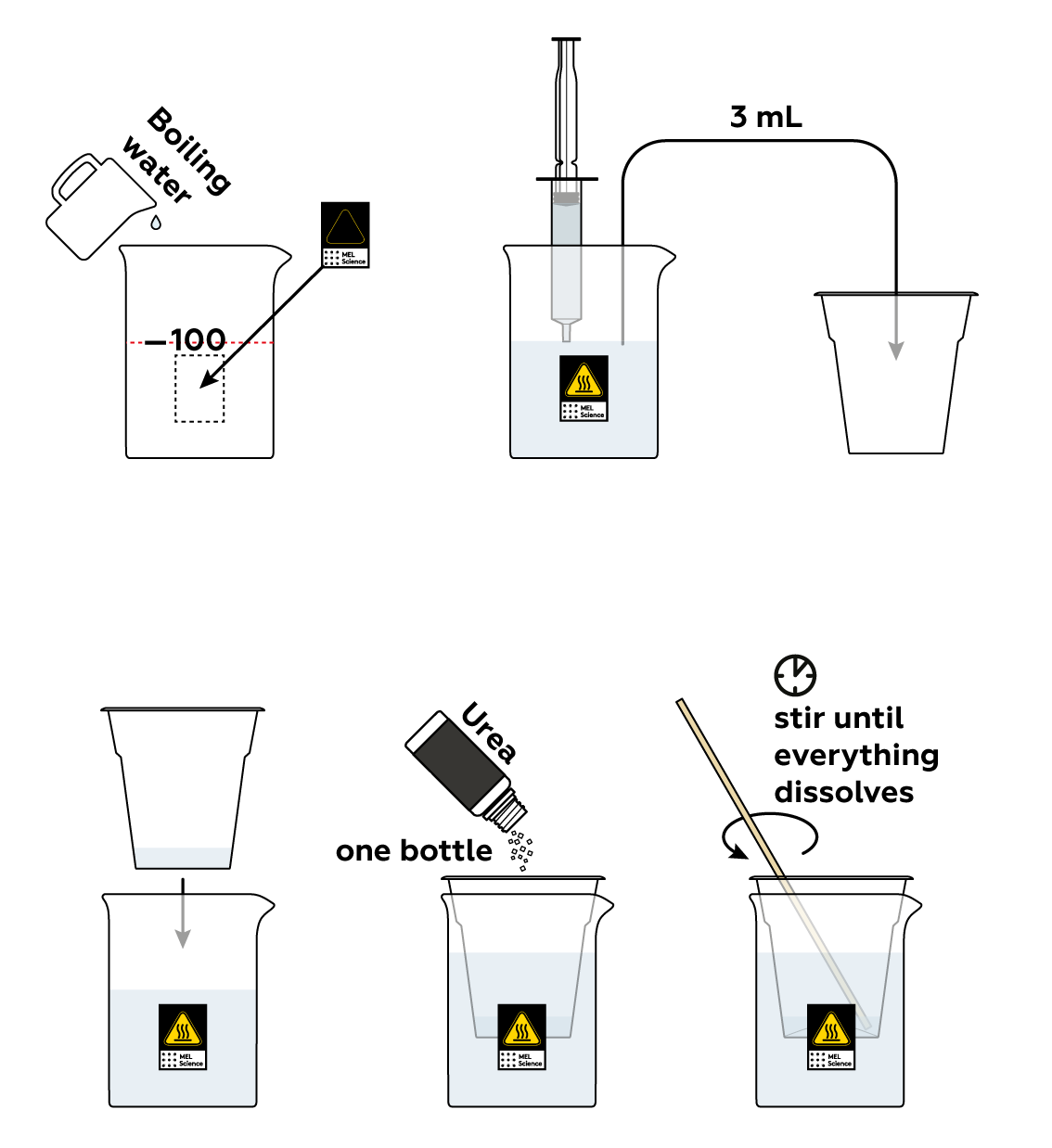
If you use too much water, your results won’t be nearly as spectacular. It’s better to start this step over. Carefully pour the water back into the cup and use a syringe to measure exactly 3 mL of hot water into the same disposable cup. Continue the experiment.
Make sure that you put the disposable cup in the beaker with boiling water. Usually, the solubility of substances increases with temperature.
It could be that the water in the beaker wasn’t hot enough (remember, this experiment calls for boiling water!). Change the water in the beaker and stir the mixture again.
Yes, everything is fine. See the next step.
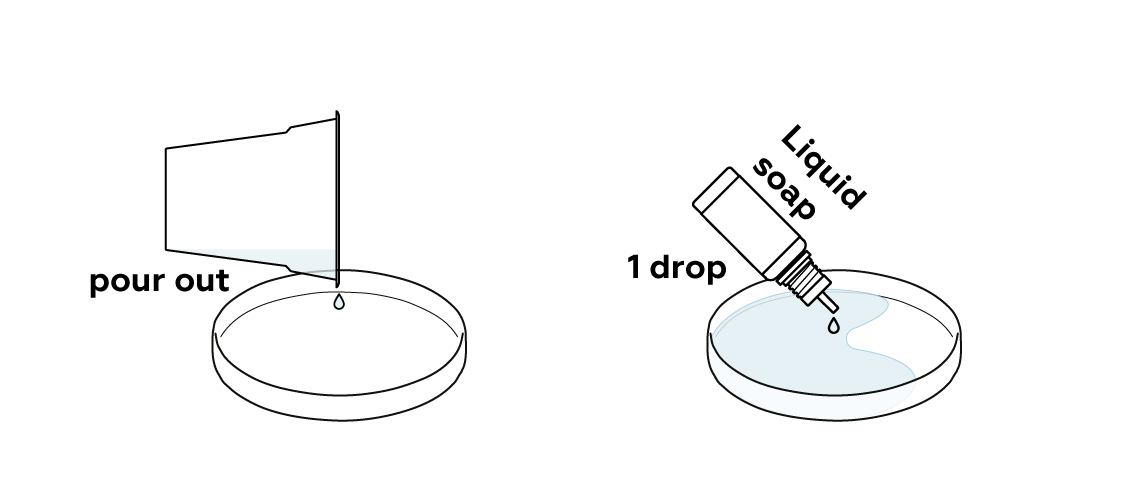
In this case, add one or two extra drops of liquid soap. Carefully swirl the Petri dish.
Unfortunately, you will have to start the experiment over.
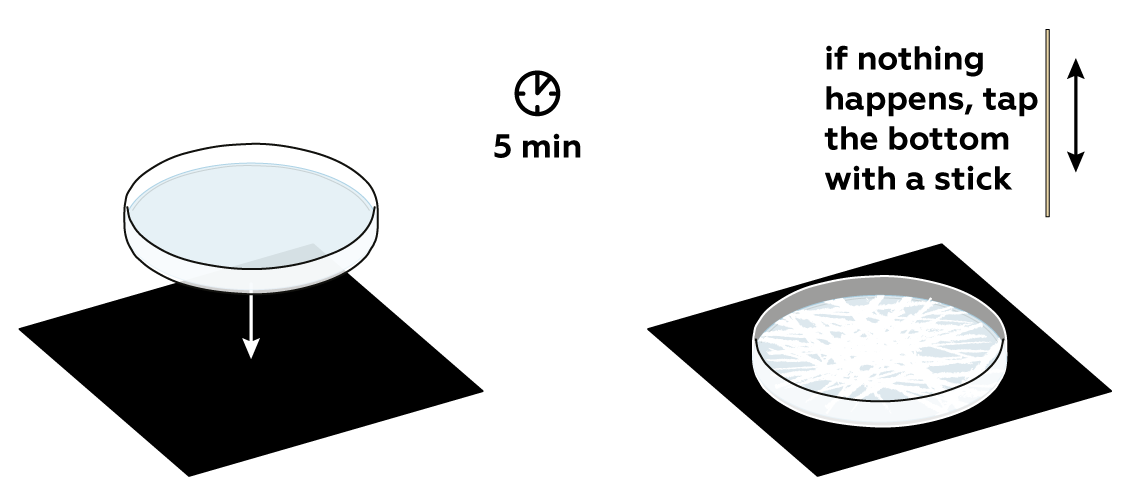
If crystals haven’t started to appear, take a wooden stick and tap the bottom of the Petri dish. Tapping puts the system out of balance, promoting the formation of centers of crystallization. This should yield beautiful crystals!
Step-by-step instructions
Many substances, urea included, dissolve much better in hot water than in cold water. So much so, in fact, that hot water can dissolve more than four times the quantity of urea cold water can.
Once you've got your hot urea solution, pour it into a Petri dish to let it cool.
As the solution cools, the solubility of the urea drops. This means that cold water can't hold the amount of urea hot water can, and urea crystals have to form, unless... If you're especially lucky, the crystals won't form on their own, and you'll get a chance to provoke their growth with a gentle tap of a stick.
Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage. Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description



around constantly. The intensity of this movement is what we call "temperature," i.e., the faster the molecules move, the hotter the substance.

, the urea molecules


.
Sugar, table salt, and most other solid substances also dissolve better when it's hot, but that's not always the case. Some substances, such as sodium sulfate Na2SO4, have a more complicated relationship with water, and their solubility can actually decrease somewhat at high temperatures.