Chemical seaweed
Grow chemical seaweed in a beaker!
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the safety underlay.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
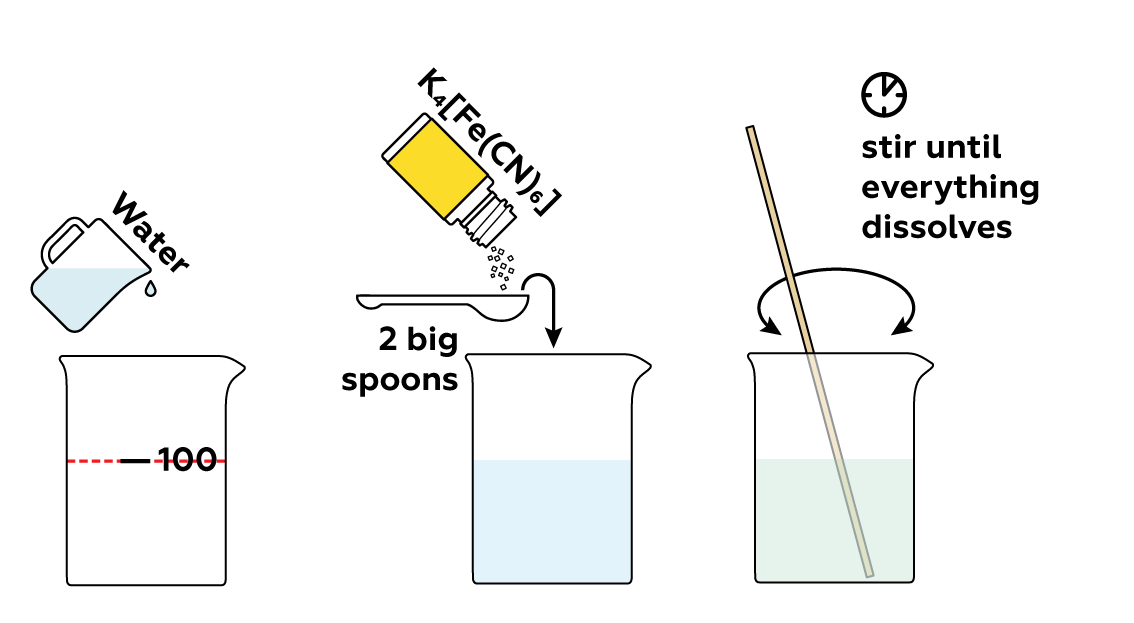
First, make sure you measured precisely 100 mL of water and the K4[Fe(CN)6] has dissolved completely. Next, make sure that there are gaps between the various salts and they haven’t mixed together in the beaker – this will hinder the seaweed’s growth. Be careful: the seaweed is very thin and fragile. Let the beaker sit undisturbed for 20–30 minutes.
If these compounds mix, it will be difficult to distinguish the different colors of seaweed. Additionally, the seaweed made from the last salt added will dominate over the rest of seaweed, and the result will not be as diverse.
Just continue the experiment and see what happens. Afterward, try repeating the experiment with larger gaps between the compounds. Compare the results. This is like an experiment within an experiment!
Step-by-step instructions
Prepare a potassium hexacyanoferrate(II) K4[Fe(CN)6] solution.
Pour various metal compounds (called metal salts) into different areas of the beaker.
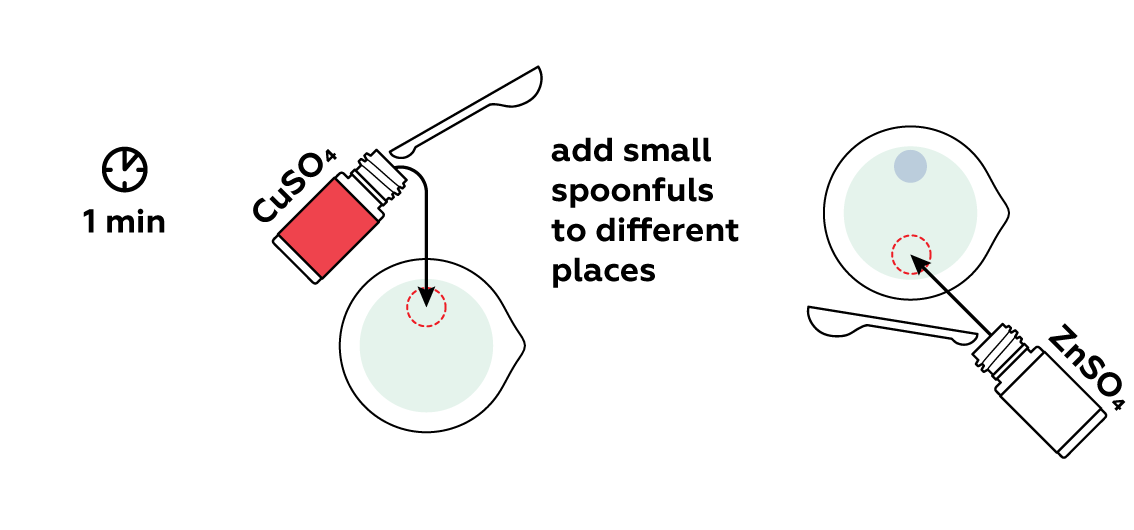
The metal salts gradually dissolve and react with the potassium hexacyanoferrate(II), forming insoluble copper and zinc compounds. These don’t just precipitate out, but rather form “bubbles” due to osmotic pressure. The elegant chemical seaweed grows from these bubbles.

A similar reaction can occur between liquids too.

Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage. Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description









. Тhе K4[Fe(CN)6] solution


, and thus the chemical seaweed grows.
Why does the chemical seaweed form?
Upon entering the solution, the salt crystals begin reacting with the yellow potassium hexacyanoferrate(II). This reaction starts on the surface of the solid salts, ultimately covering each of the crystals with a film of insoluble compounds. When this happens, the reaction is momentarily "halted." But only momentarily – potassium hexacyanoferrate(II) seeps through the gaps in the insoluble coating, and the reaction begins again. It halts once again as a new layer of insoluble compounds forms. Potassium hexacyanoferrate(II) breaks through the coating once more. And so on and so forth, lengthening the chain of compounds onwards and upwards. And thus the fascinating structures at the bottom of the beaker come to be.
Why does the seaweed vary in appearance?
The obvious guess would be that the different substances – white zinc sulfate ZnSO4 and blue copper sulfate CuSO4 – “grow” differently. This is actually pretty close to the right answer.
One the one hand, these compounds are quite similar – they are all metal sulfates. They consist of a positively-charged metal ion and a negatively-charged sulfate ion, which they break down (dissociate) into in water:
ZnSO4 ↔ Zn2+ + SO42- CuSO4 ↔ Cu2+ + SO42-
On the other hand, they differ in color and appearance, one being a fine white powder, the other being bluish crystals.
Ultimately, it’s the metal ion that gives each sulfate its properties and makes each substance react with the (K4[Fe(CN)6]) solution in its own unique manner.
Why does the copper sulfate change colors in the solution?
You may have noticed that the initially-blue copper sulfate CuSO4 changes distinctly during the experiment. The “seaweed’s” reddish-brown color is a result of the formation of potassium-copper ferricyanide:
CuSO4 + K4[Fe(CN)6] → (K2Cu[Fe(CN)6])↓ + K2SO4
How can you make the seaweed grow differently?
To alter how the seaweed grows, try adjusting the parameters of the experiment. More precisely, you can try adapting the parameters of the beaker, the volumes of all the solutions, and the amount of crystals used. For example, you can use a more concentrated solution of potassium hexacyanoferrate(II) (just add less water, 50 mL instead of 100 mL). This is a tried and tested method.
Ultimately, don’t be scared to experiment on your own! Try using hot or cold water, or pick out the smaller/bigger crystals. Make sure to observe the proper safety precautions when working with chemical compounds, and do write down what conditions you changed in order to compare your results and draw conclusions. This is pretty much the life of a modern-day scientist!
Why shouldn’t you shake the beaker during the experiment?
The forming structures are just heaps of insoluble crystals accumulating rather chaotically, and so are quite fragile. Even the slightest disturbance can transform your artificial seaweed into a murky brew. You can test this quite easily – just prod the seaweed slightly with something like a toothpick or a match.