Acidic patterns
See how thymol blue reacts to changes in pH!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the safety underlay.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
Add just as much water as needed to cover the bottom of the Petri dish. Be careful not to add too much – you’ll mix the contents of the Petri dish as part of the next step.
Add a few more drops of Na2CO3 solution and stir.
Don’t worry, this isn’t critical. Just continue the experiment.
You can use various water-soluble substances of varying acidity to create patterns just like in our experiment. For example, a solution of regular baking soda has an alkaline pH (i.e. the solution is basic). When making this solution, use hot water to help the baking soda dissolve more easily.
You can also use smelling salts which, when dissolved in water, form an ammonia solution, which is also a basic medium. However, this solution exudes an aggressive odor, so it’s best to work with it in a well-ventilated area or even outside.
Meanwhile, you can use solutions of citric or acetic acid or add some lemon juice to serve as acidic media.
Step-by-step instructions
Prepare a diluted sodium carbonate solution. This is an alkaline solution—its pH is higher than neutral.
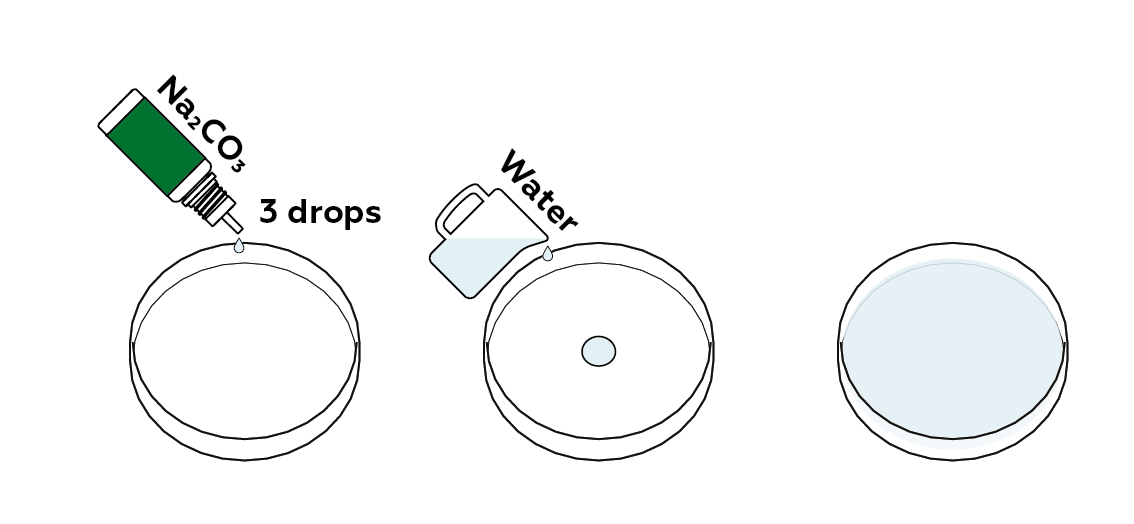
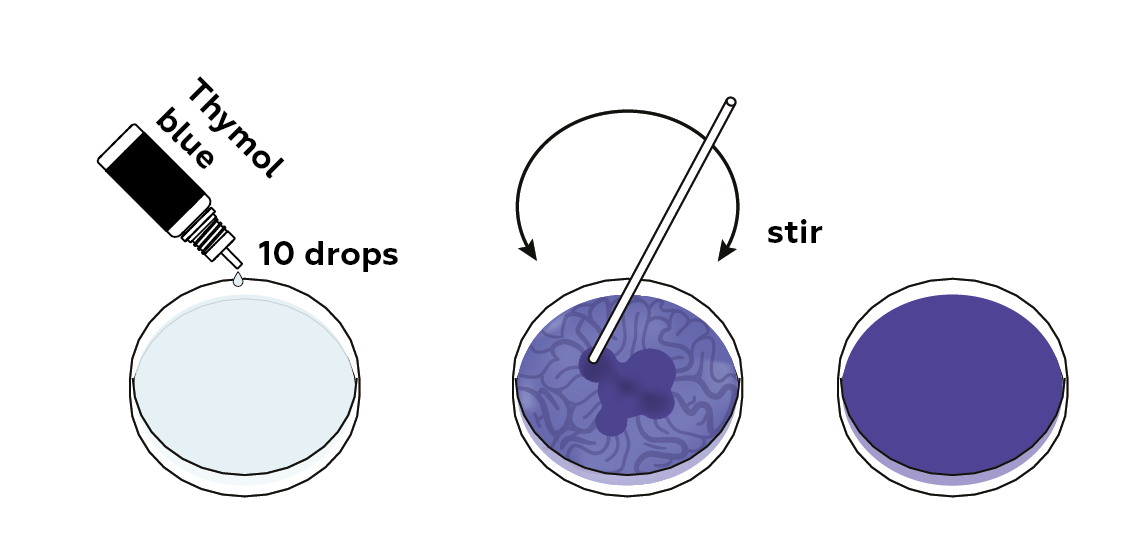
Add the pH indicator, thymol blue. It will turn blue in an alkaline medium.
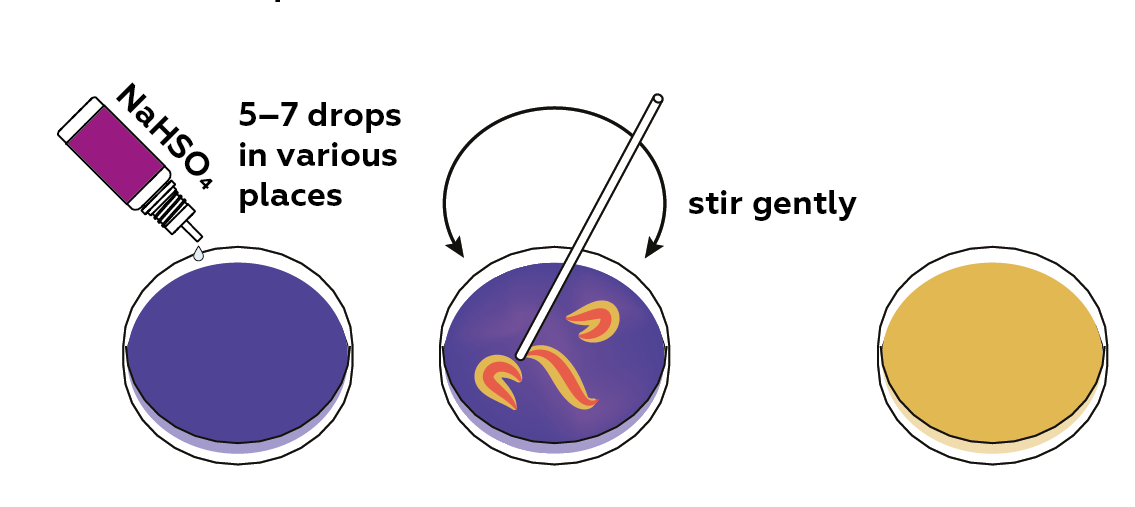
Now, add sodium hydrogen sulfate NaHSO4 (an acid salt) to lower the pH of the solution.
Now, add sodium carbonate Na2CO3 (an alkaline solution) to raise the pH of the solution.
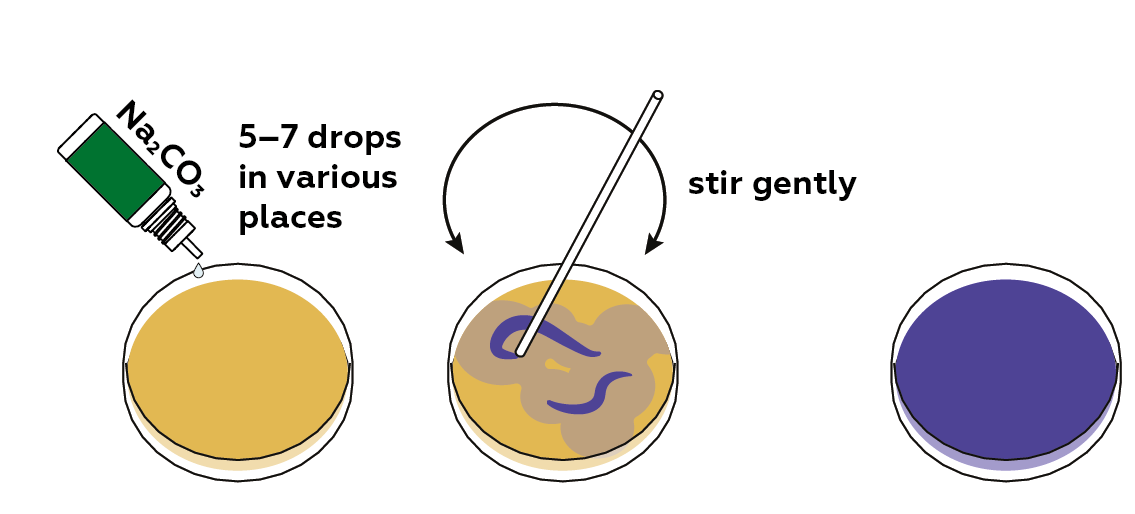
You can repeat steps 3 and 4 several times. Try adding varying volumes of NaHSO4 and Na2CO3.
Expected result
Thymol blue turns blue when added to a basic medium. If an acidic solution of sodium hydrogen sulfate is added, and the mixture is stirred gently, orange patterns will appear in the Petri dish.
Disposal
Please refer to local regulations when disposing of chemicals. Dispose of other solid waste with household garbage. Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description


ions in the solution, raising the solution’s pH. Sodium carbonate Na2CO3 is one such substance.
When combined, H+ 
make water H2O
. This process is called a neutralization reaction. We can use our pH indicator to observe how the acidic medium (low pH) created by NaHSO4 is neutralized by the basic medium (high pH) of Na2CO3, and vice versa.

Why does thymol blue change color?
Thymol blue changes color depending on the medium it is in. In other words, it is a pH indicator. In a basic medium, it turns blue; in an acidic medium, it turns red; in an almost neutral medium, it turns yellow.
Blue, red, and yellow are considered ‘primary’ colors because combining them can yield many additional variations: blue + yellow = green, yellow + red = orange, red + blue = violet. You can create these combinations in your Petri dish too! Just stir the contents gently with a wooden stick.
How does the acidity of the solution in the Petri dish change?
The acidity of a solution depends on its ratio of positively-charged hydrogen ions H+ (protons) to negatively-charged hydroxide ions OH-. A solution is acidic if it contains more H+ ions than OH- ions; conversely, it is basic if it contains more OH- than H+.
Ordinary water is practically neutral: the numbers of H+ and OH- in it are almost equal. However, some substances can increase the concentration of H+ or OH- ions, thus increasing or decreasing the pH respectively.
For example, sodium carbonate, which we added to the Petri dish first, creates a basic medium. Its interaction with water produces a large amount of hydroxide ions OH-:
Na2CO3 + H2O → 2Na+ + HCO3- + OH-
This abundance of hydroxide ions makes the solution in the Petri dish basic. Thymol blue can be used to confirm this – it turns blue when added to the solution.
Sodium hydrogen sulfate, in contrast to sodium carbonate, dissolves in water and releases protons H+:
NaHSO4 → Na+ + H+ + SO42-
The solution becomes acidic as soon as sodium hydrogen sulfate is added to the Petri dish.
As we add reagents to different places in the Petri dish, the solution’s acidity changes unevenly. In some places, H+ ions dominate, while OH- reign in others. As a result, the thymol blue peppers the solution with a variety of colors. When mixed, the acidity of the medium becomes uniform all over the solution, so the thymol blue’s color becomes uniform as well.
That’s interesting!
How do pH levels affect living organisms?
Changes in the pH of various solutions around or even inside living organisms can strongly affect their quality of life. For example, a pH below 6 is usually too acidic for fish. Even if it won’t kill fish and other aquatic species outright, it can interfere with their growth and reproduction. Fish eggs will not hatch if the pH of the water is below 5. On the other hand, if the pH rises above 9, fishes’ ability to absorb oxygen worsens, and they can even suffocate.
It is also important to monitor the pH of rainwater, which can be quite acidic due to the presence of sulfuric acid H2SO4 and nitric acid HNO3. These acids form in the atmosphere from nitrogen and sulfur oxides, which are emitted as waste by numerous industries, transport systems, boilers, and thermal power plants. Acid rain with a low pH (less than 5.6) destroys flora and sea life. Thereafter, in acidic soil, trees and plants are less able to absorb the groundwater they need to grow.
pH is also important for human beings, as the body maintains a certain acid-base balance. Disturbances in this balance can lead to many serious ailments. Each individual organ or bodily fluid (such as blood or saliva) maintains a certain pH. The stomach, for instance, houses an extremely acidic medium (pH 1.8 – 3.0). This medium is maintained by hydrochloric acid HCl, which promotes the breakdown of food in the stomach and acts as an antibacterial agent. The digestive products then enter the intestine, where the pH is alkaline (pH 7 – 8). Thus, acid is formed in one area of the stomach and neutralized in the other (at the bottom of the stomach):
HCl + NaHCO3 → NaCl + CO2 + H2O
Stomach acid is mainly neutralized by sodium bicarbonate NaHCO3.
