Quantitative analysis
Let’s determine chloride ion content in water!


Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the tray.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
What drinking water to use for the experiment?
Bottled drinking, still mineral or simply boiled water would work for this experiment.
After adding silver nitrate AgNO3 solution, the water is so turbid that the scale is not visible. What to do?
It looks like there is a very large content of chloride ions in the water you have analyzed. To conduct its analysis, it is necessary to calibrate again. In step 2, add 2-3 drops (instead of 5) of silver nitrate. Follow the instructions to step 9. In step 9, also add 2-3 drops AgNO3. Please note that in steps 2 and 9, you must ensure that strictly the same amount of silver nitrate is added. Complete the experiment following the remaining counts in the instructions.
How to determine chloride Cl- concentration in drinking water?
Mixing sodium chloride NaCl with silver nitrate AgNO3 results in precipitation of insoluble silver chloride AgCl. The more of it forms, the cloudier it makes the solution. In this experiment, we add an excess of silver nitrate, so that the amount of precipitate formed depends solely on the amount of sodium chloride used. In terms of chloride anions, each drop of sodium chloride solution adds 40 mg of anions per 1 L of water. Therefore, in order to calculate the concentration of chloride ions in water, we should multiply the quantity of drops, which have been added in step 11, by 40.
But how did we figure out that one drop of sodium chloride provides for 40 mg/L of chloride anions? Let’s see:
• For this experiment, we took 0.1M sodium chloride solution. Thus, when we add one drop of it (i.e. 0.3 mL) to 3 mL of distilled water, it is being diluted 90-fold to a 0.0011M solution.
• Molar mass of sodium chloride is 35.5 g/mol.
• Finally, we calculate the concentration of chloride ions: 0.0011М * 35.5 g/mol = 0.0391 g/L, or 39 mg/L. Round off the number, and the result is 40 mg/L of chloride ions.
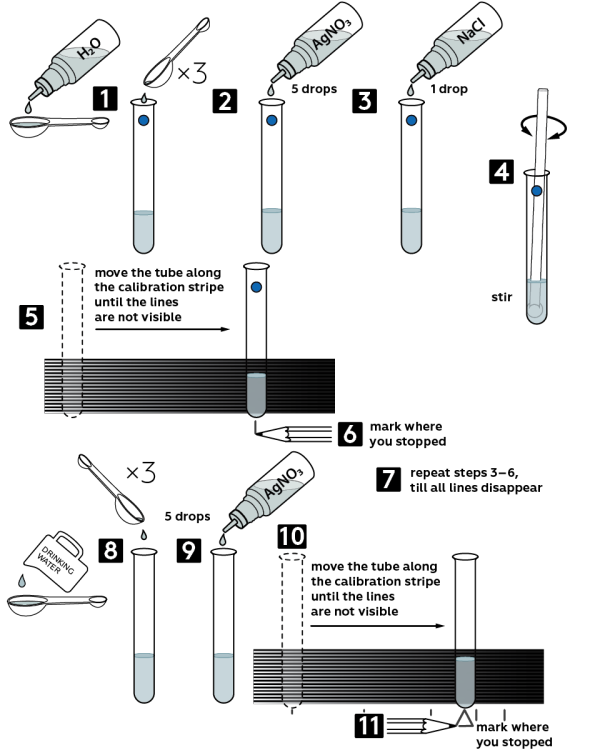
Step-by-step instructions
- Take a test tube marked with a blue circle and pour in 3 big measuring spoons of distilled water.
- Add there 5 drops of 0.5M silver nitrate AgNO3 solution.
- Add 1 drop of 0.1M sodium chloride NaCl solution.
- Mix the contents with a plastic stirring rod.
- Take a turbidity standard. Starting from the black-white end of the standard, move the test tube along until the stripes cannot be distinguished anymore.
- Mark the place where you stopped.
- Repeat steps 3−6, each time until the stripes cannot be seen.
- Take another test tube and pour in 3 big measuring spoons of drinking water.
- Add there 5 drops of 0.5M silver nitrate AgNO3 solution.
- Starting from the black-white end of the standard, move the test tube along until the stripes cannot be distinguished anymore.
- Mark the place where you stopped.
Disposal
Dispose of solid waste together with household garbage. Pour solutions down the sink. Wash off with excess of water.
Scientific description
How do chlorides in water influence our health?
Chloride anion Cl- can be considered the most important anion in human body. Its value is difficult to overestimate. First, it participates in food digestion process because of hydrochloric acid HCl in our stomach. In aqueous solution, it exists in form of two ions: H+ and Cl-. Second, chlorides maintain certain pressure in cells of our body. Third, chlorides contribute to transmission of nerve impulses. Unfortunately, though, an excessive intake of chloride anions is harmful for human, too. Then, what to do with chlorides in water?
In most countries, the maximum permissible concentration (MPC) of chlorides in water is 300–350 mg/L. Human sense of taste recognizes even lower concentrations in water – as a salty taste. Thus, water that isn’t salty surely doesn’t contain too much chlorides.
Nevertheless, drinking a lot of mineral water with high chloride ions content may lead to fluid retention and, as a result, to excessive liquid accumulation in the body. Notably, the reason of such swelling is not chloride ions but sodium ions that always accompany them since the main (and almost the only) source of chlorides is table salt NaCl.
We should also mention one interesting but unpleasant fact, which is true for almost any populated area. If drinking water suddenly becomes salty, it probably means that a water-bearing deposit or a waterline has been polluted with sewage water. In fact, human and animal waste products are rich in chlorides.
Seawater contains significant amount of salts, including chlorides, which makes it undrinkable. In general, one liter of seawater contains 19 grams of chlorides – it is 54 times higher than the maximum permissible concentration for drinking water!
Chloride ions deficiency in drinking water could hardly be harmful for health of someone who cares about diverse nutrition. The main sources for chlorides are meat, fish, seafood, diary, and seaweeds. Moreover, chlorides are also found in berries, fruits, and sweets!