Neon atom
An interactive lab where students assemble their own neon atom with the first two complete electron shells.
This lesson is a part of MEL VR Science Simulations. Learn more →
Similar lessons
Transcript



Teacher's notes
Keywords
neon, atom, nucleus, proton, neutron, isotope, electron, electron orbital, electron configuration diagram, s-orbital, p-orbital
Students will
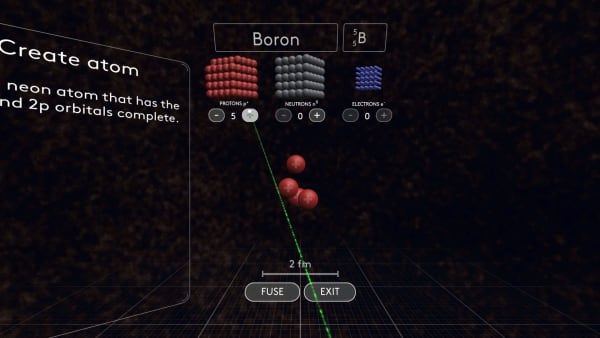
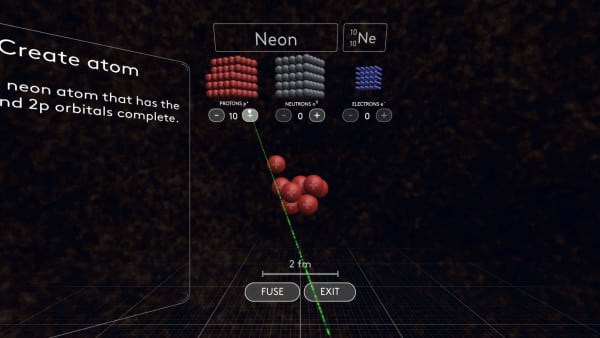
- Build a neon atom knowing the number of fully-filled orbitals
- Review the number of electrons in fully-filled orbitals and the number of orbitals
- Review electroneutrality and calculate how many protons a neon atom has
- Calculate how many neutrons a neon atom has
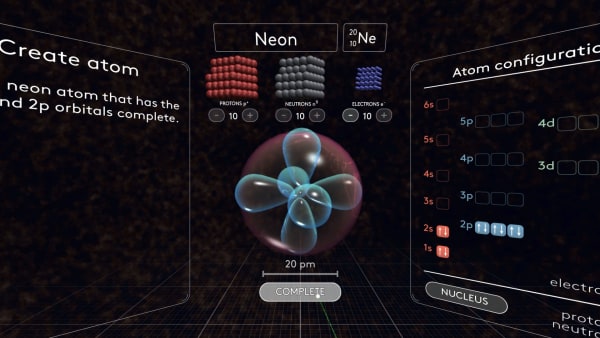
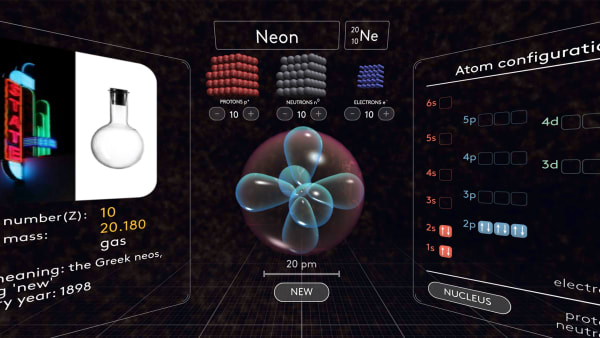
- Review electron orbital names, shapes, and order of appearance
- Review electron configuration diagrams
The lab primarily serves as a skill builder for electron configuration. It can be used in a variety of topics throughout the curriculum: periodicity and properties, ion formation.
Topics to discuss
- Noble gases
- Electron shells



