Diamonds – shining and ancient
How the “unbreakable” mineral was created
Diamonds as jewelry have been known to humanity for a long time – 5,000 years. This mineral stood out from others for its unique beauty, and for centuries it was a sign of wealth. Let’s take a closer look at them.
What is a diamond?
The word “diamond” comes from the Ancient Greek “adamas”, which means “unbreakable”. Indeed, in ordinary conditions, the mineral can maintain a stable state for an unlimited amount of time. Diamonds are the hardest but also the most fragile mineral known to humanity. The mineral is used in various fields of industry because of its hardness (diamond circles on metal, use in electronics). An interesting fact is that most diamond jewels have a yellow or brown tint. People have even learned to make artificial diamonds. They are made and elaborated under colossal pressure.

Diamonds are a rare mineral, but can be found all over the world. Diamond mines can be found everywhere but Antarctica. Scientists are still puzzled by the formation of the mineral – what natural phenomenon causes diamonds to form?
A simple question which is difficult to grasp: how can a diamond burn?
Indeed, is this actually possible? You can’t just take the hardest mineral on Earth and burn it. But even if you could, why would you want to? Let’s look at several discoveries which were made in the past.
Through the ages
Before the 17th century, diamonds were regarded as a substance that had nothing in common with graphite or coal. The scientist Axel de Brut, and later Isaac Newton, believed that the mineral could burn. They came to this conclusion from observations of the refractions of rays of turpentine and camphor.
In 1694, Florentine scientists conducted an experiment to find out what would happen if they put a diamond in the focus of a concave mirror. They achieved the desired result – the diamond began to smoldered, and after further heating, the theory became reality – the diamond burnt down completely.
Scientists at the time did not draw the correct conclusions from the experiment, as they believed that diamond and quartz were the same substance, with different types of internal structures.
In 1772, Antoine Lavoisier held a similar experiment to show the scientific community that the theory was correct. Creating the conditions for the experiment did not take him much time or effort – he only required a large lens, a sealed boiler and the help of several assistants. Lavoisier’s opponents hoped that he would fail, but the experiment was a success. The stone disappeared from the effect of the sun’s rays, which made Lavoisier conclude that it had turned to gas.
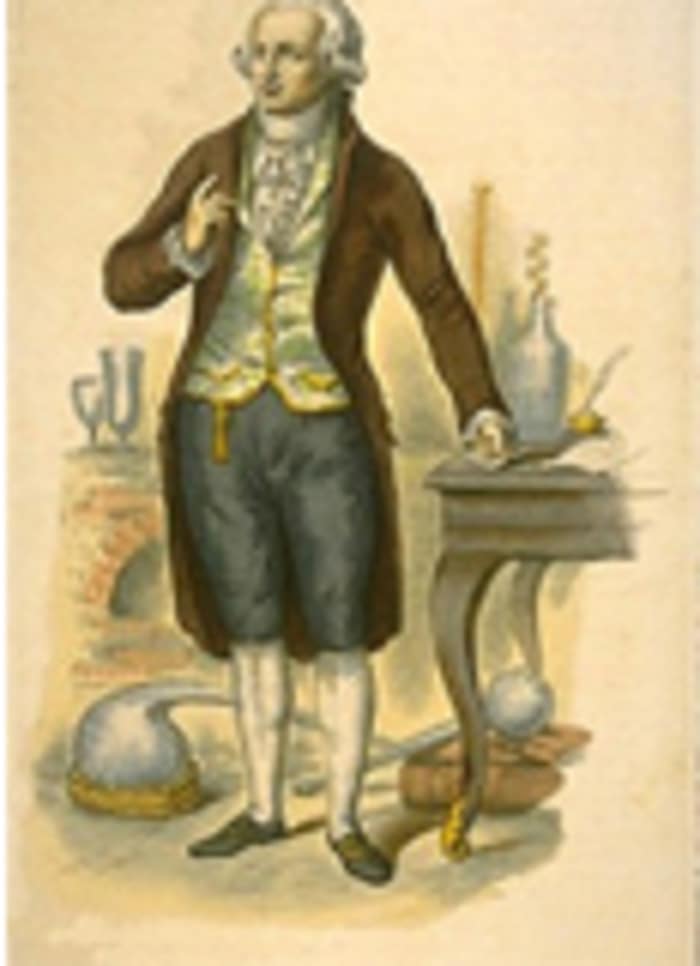
In 1797, Smithson Tennat made a similar experiment. He took a gold pot with a pipe attached to it, and oxygen was pumped into the pot. According to his calculation, by gradually adding oxygen, the mineral would transform into a gaseous state, i.e. the diamond would turn to gas, just like when coal was burned. The experiment was a success, as it had been for Lavoisier. The gas in the pot was carbon dioxide, which confirmed his suppositions. The theory worked.
The scientist De Morveau also conducted studies in this field. In his experiments, the scientist also decided to burn graphite, which had a carbon content of over 99.5%. De Morveau found an interesting correspondence – if identical conditions were created for burning graphite, diamond and coal of identical weight, then the same amount of carbon dioxide would form. Here you'll find easy and safe experiments on obtaining beautiful minerals.
Just carbon
We may often wonder why carbon dioxide is formed when diamonds are burnt. The explanation is simple: diamonds are an allotropic form (“structural modification”) of carbon. Here is diamond’s crystal structure:
Indeed, diamonds burn in the open air at a temperature of 850-1000 °С, and in pure oxygen at 750 °С.
A final word about safety
Remember that the experiments described in this article were conducted by professionals in the field of chemistry, even if this did take place a long time ago. We don’t recommend repeating them just to find out what the result will be. We strongly recommend that you refrain from conducting experiments of this kind yourself, as there will be unforeseen and unpleasant consequences.
