Metal contest
Figure out which metal is the most reactive!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
First, try using another bottle nozzle. It should fit snugly in the bottle. And, of course, you can ask an adult for help.
Yes, of course, you can. But be sure to take a cap from a bottle meant for liquids! Otherwise, you won’t be able to close the bottle tight enough.
Don't worry! Just continue the experiment.
Yes, the reaction is proceeding, but slowly: metallic copper is accumulating on the surface of the zinc wire in the form of a dark precipitate.
No, unfortunately, you will not be able to keep the needles – they are extremely thin and fragile. They will immediately stick together if you try to remove them from the solution.
Step-by-step instructions
Prepare a copper sulfate CuSO4 solution.

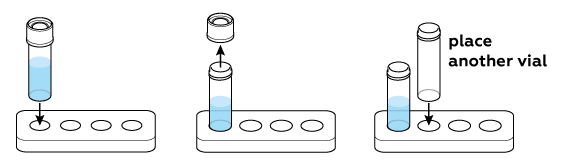
Arrange the vials in your stand.
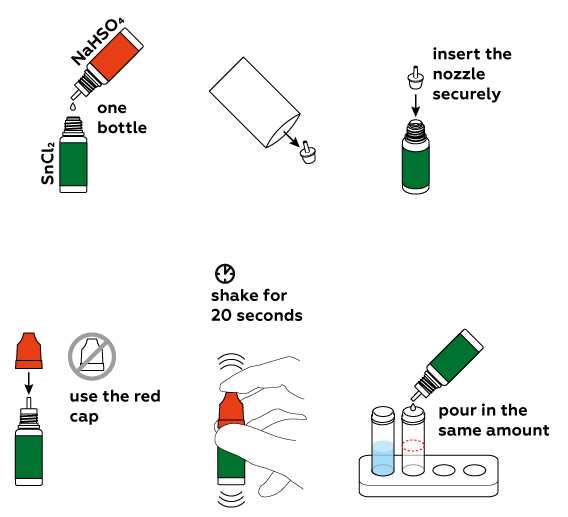
Prepare a tin chloride SnCl2 solution.
Put pieces of zinc into both vials.
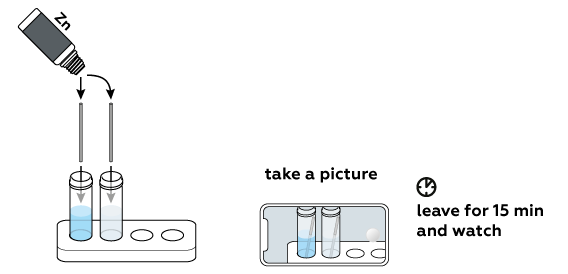
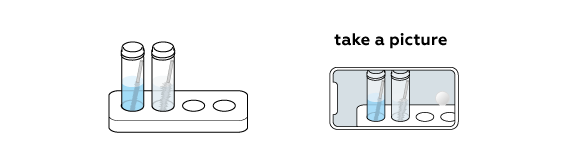
Look for signs of a chemical reaction!
Disposal
Dispose of solid waste along with household garbage. Pour solutions down the sink. Wash with an excess of water.
Scientific description

inside a cloud of electrons
. Ions of some metals value the comfort of this cloud more than others. They can push less lazy metals away


If you arrange metals from more active to less active, you'll see that zinc Zn is a more adventurous fellow than tin Sn and copper Cu. That's why, when you put a zinc rod into a solution containing, say, copper ions Cu2+, the latter are happy to settle inside the comfortable cloud of electrons, forming metallic copper Cu on the surface of the rod. Zn ions Zn2+, in turn, go swimming in the solution. The reaction with the SnCl2 solution is essentially the same.

What is the “metal reactivity” series?
Let’s start with an analogy. A sports competition is a contest to determine the strongest athlete or team, where athletes compete with one another to find out who is who. This usually results in a scoreboard showing the names of the competitors from strongest to weakest.
The same holds true for metals. Some metals are more active than others. But unlike athletes, who can develop their skills and improve their ranking position on the scoreboard, metals have no such opportunity. For example, zinc will always be more active than copper – the opposite will never be true. Chemists were able to create a league table of metals (like a sports competition scoreboard) known as the metal reactivity series. The most active metals are on the left side of the table, while the least active ones are on the right:
Li → Rb → K → Ba → Sr → Ca → Mg → Al → Mn → Cr → Zn → Fe → Cd → Co → Ni → Sn → Pb → H → Sb → Bi → Cu → Hg → Ag → Pd → Pt → Au
How does the metal reactivity series work?
In our experiment, we put a piece of zinc wire into a solution of copper sulfate. We then watched a deposit of metallic copper form on the piece of zinc according to the reaction:
Zn + CuSO4 → Cu + ZnSO4
In this case, zinc literally forces copper out of its salt composition. The same happens in the vial with the zinc wire and the tin chloride solution, where zinc takes tin’s place:
Zn + SnCl2 → Sn + ZnCl2
In the vial with the tin wire in the copper sulfate solution, the reaction proceeds more slowly, but still surely, and the tin is slowly covered with a thin layer of copper. This reaction gradually pushes the copper out of the copper sulfate solution and onto the tin.
Sn + CuSO4 → Cu + SnSO4
As per the metal reactivity series, zinc is more active than both tin and copper, while tin is more active than copper. This is how the metal reactivity series works: more active metals push less active metals from their salts, but never vice versa. This is why we did not observe any reactions in the rest of the vials.
Hydrogen is not a metal. Why is it included in the reactivity series?
Hydrogen helps arrange the metals according to their activity. The metals to the right of hydrogen are relatively nonreactive. For example, they will not react if put in an acidic solution.
The metals in the middle of the reactivity series, close to the left of hydrogen, have medium reactivity. These metals react with the acid in an acidic solution, producing hydrogen as a product. Zinc is an example of such a metal. The following reaction shows how zinc reacts with a sulfuric acid solution:
Zn + H2SO4 → ZnSO4 + H2↑
Alkali metals and some alkaline earth metals react differently from other metals. When put in an acidic solution, they react with water. As such metals are the most reactive, they are located at the very beginning of the reactivity series.
