Burning sugar
Sugar doesn't burn easily. Simply add ash to set it alight.


Reagents
Safety
-
Put protective eyewear on.
-
Conduct the experiment on the tray.
-
Keep a bowl of water nearby during the experiment.
-
Place the stove on the cork hot pot stand. Do not touch the stove after the experiment; wait until it gets cold.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
What if the sugar still doesn't burn after ash is added?
Make sure that you rub ash onto each face of the sugar cube, and, when you place the sugar cube on top of the other sugar cube, turn it a bit so that the corners stick out. This offset should make it easier to ignite the sugar. Try to ignite the sugar with a lighter instead of a match.
Step-by-step instructions
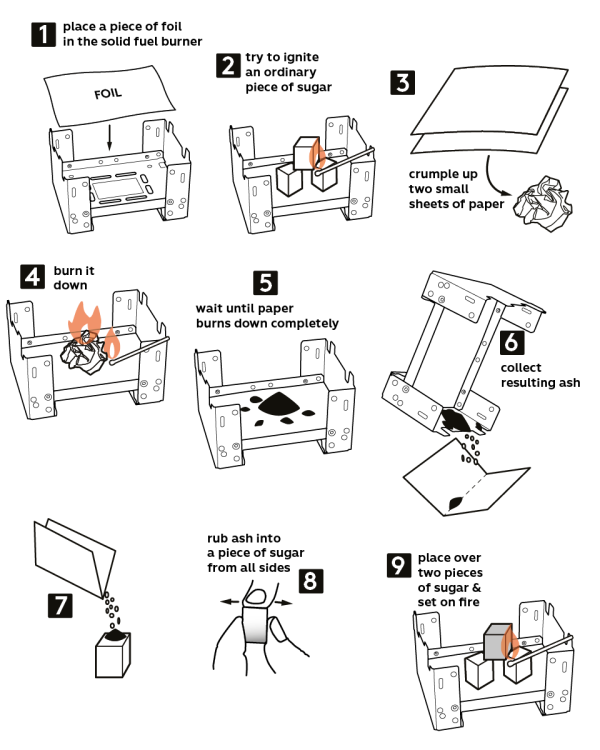
-
Take the solid fuel stove from the Starter kit. Place a piece of aluminum foil onto the stove. Caution! Use a cork hot pad to protect the tabletop.
-
Make a “pyramid” from three sugar cubes, as shown. Try igniting a regular piece of sugar – it won’t burn.
-
Crumple up two pieces of paper and place them onto the stove.
-
Now, burn that crumpled paper.
-
Wait for paper to burn completely.
-
Collect ashes.
-
Pour the ash on top of a sugar cube.
-
Rub ash into the sugar cube, from all the sides.
-
Finally, put that sugar cube on top of the other two (as shown) and ignite it. Perhaps, it would need some time to ignite.
Expected result
Sugar typically does not burn, but add ash and the sugar cube will ignite.
Disposal
Dispose of solid waste together with household garbage.
Scientific description
Why do some substances burn and some do not?
Let's look at the process of burning and exactly what it is. Burning is the oxidation reaction of a substance by oxygen. Oxygen takes away electrons from a substance, breaks the bonds between atoms in the substance and inserts itself into the product molecules, which results in a mixture of oxides. The difference between burning and other oxidation reactions (e.g. iron corrosion) is that large amounts of heat are released during burning. There are two conditions needed for a substance to burn:
- During oxidation the amount of heat released must be sufficient to sustain burning
- The oxidation reaction must be quite fast
Why doesn't the sugar cube burn?
The oxidation of sugar is capable of releasing a lot of heat. If you look at the nutrition information on a pack of cookies, you can see that they contain high levels of sugar and calories. The number of calories in food equates to the energy generated during their oxidation/digestion in the body. However, the oxidation of sugar is quite slow, so sugar does not burn easily.
Why does the sugar cube burn when you coat it in ash?
Ash serves as a catalyst (accelerator) in the burning reaction of sugar. A catalyst is a substance that increases the rate of a reaction, by lowering the amount of energy needed for the reaction to work.
What is ash? What is it made of?
Ash is a complex mixture of substances that does not continue to burn (be oxidized), which contains salts or oxides of metals. Ash from paper contains metal carbonates, such as salts of carbonic acid (H2CO3). These salts or oxides act as catalysts in the sugar burning reaction. The best catalysts are lithium salts, which are found, for example, in cigarette ash. However, paper ash contains sodium and calcium salts that can also catalyze the combustion of sugar.
What is left over from burning sugar?
If sugar burns completely (complete combustion) then the products of the reaction are carbon dioxide and water. However, in reality, sugar tends not to burn completely and a black mass of products of incomplete oxidation of sugar result. The black mass contains complex organic substances (aldehydes, ketones, carboxylic acids) and carbon.
That’s interesting!
Why does burning sugar smell?
When the sugar decomposes with heat, a compound called maltol is produced, which has a characteristic "caramel" smell of burning sugar. Maltol is a naturally occurring compound that is often used as a flavor enhancer, and contains a ketone (a double bond from carbon to oxygen) and an alcohol (a single bond from carbon to OH) group.
