Properties of mercury oxide and the reaction of its breakdown
Obtaining mercury oxide illustrated by an experiment

Mercury oxide is a binary compound of oxygen and mercury, with the formula HgO. In normal conditions it is a solid, loose substance, and depending on the degree of dispersion it is red or yellow – the main and most important mercury oxide. It is hardly ever found in nature, one exception being the rare mineral of montroydite. Using mercury oxide, in 1774 the scientist Joseph Priestley discovered oxygen (the reaction of the breakdown of mercury oxide).
Properties of mercury oxide
Yellow HgO is a more chemically active substance, which breaks down at a temperature of 332 °С, and turns red when heated. Red mercury oxide breaks down at a temperature of 500 °С, and when heated it changes its color to black (reversible reaction).
Mercury oxide (II) is poorly soluble in water, and the substance shows weak base properties. It dissolves in concentrated solutions of alkalis, forming hydroxocomplexes. Yellow HgO interacts with NH₃ with the formation of Millon’s base, and the reaction of the equation is
2HgO + NH₃ → [Hg₂N]OH · H₂O + Q
This substance enters into a reaction with acids, forming corresponding salts. It is used to obtain mercury, and also in some types of electric cells. Mercury oxide has a strong toxic effect.
Obtaining mercury oxide illustrated by an experiment
Mercury oxide (II) is a useful reagent, which can be used in the laboratory to obtain various mercury salts – for example mercury chloride or acetate (II). Mercury acetate (II) is used in organic synthesis, for example for obtaining aluminum isopropylate, and with HgCl₂ active magnesium amalgam can be obtained.
To conduct the experiment the following equipment is required:
- a flask with a ground glass joint;
- test tube;
- reflux condenser;
- a fritted glass filter;
- a conic flask.
Reagents used:
- nitric acid 65%;
- mercury;
- caustic soda;
- sodium chloride or hydrochloric acid
Safety rules for the experiment
As nitric oxides (II) and (IV) are poisonous and have a carcinogenic effect, caution must be observed. Mercury salts are toxic for human beings, and also dangerous for the environment. Poisonous mercury nitrate can easily be absorbed through the skin. You must work with a fume hood and a reflux condenser, as the released gases often contain mercury fumes, which are dangerous in themselves. The synthesis should be carried out with extreme caution. A fatal dose of mercury nitrate is from 0.2 to 0.4 gr. Here you’ll find safe chemistry experiments to do at home.

Process of synthesis of mercury oxide
Suspend 30 g (0.15 of a mole) of mercury in a test tube. Into a flask of 250 ml with a reflux condenser, pour 60 ml (0.9 of a mole) of HNO₃. Add mercury to the acid in small portions with a pipette, and the reaction will take place. After all the mercury is added, the reflux condenser is put on again. The solution heats and “boils” from the vigorous release of nitrogen dioxide. As the reaction ends, the release of brown gas stops, and the solution in the flask becomes colorless. Equation of the reaction:
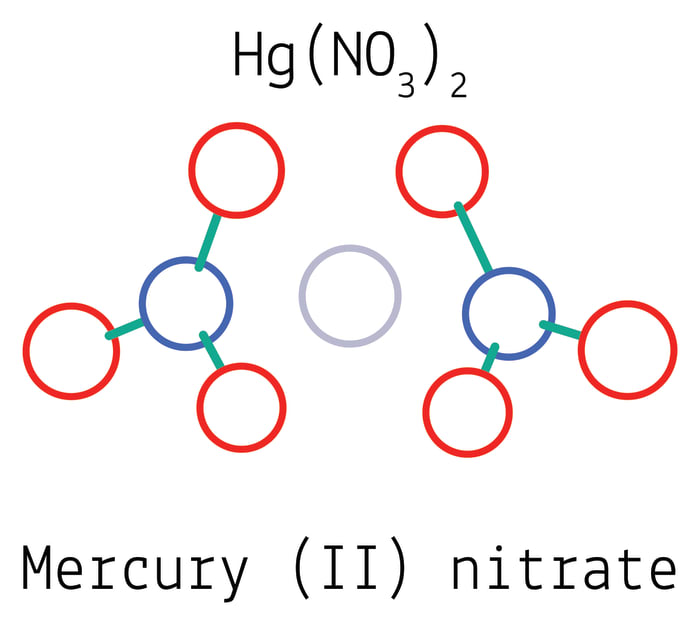
Hg + 4HNO₃ => Hg(NO₃)₂ + 2NO₂ + 2H₂O
Nitric acid is used in abundance to avoid the formation of mercury nitrate (I). The liquid cools, and HCl or NaCl is added to it – this is a test for the presence of mercury (I) – Hg₂²⁺. When a sediment of Hg₂Cl₂ settles in the solution, mercury (I) is present. A little nitric acid must be added to the solution, then it must be heated. If the test for the presence of mercury (I) is negative, then 250 ml of 4 M sodium hydroxide is slowly added to the solution. An orange sediment of mercury oxide (II) HgO forms, which is filtered. The equation of the reaction:
Hg(NO₃)₂ + 2NaOH => HgO + 2NaNO₃ + H₂O
The product is rinsed with water in the filter and dried to a constant mass in a dessicator over silica gel. The release of mercury oxide (II) is 32.467 g.
Safety rules for conducting experiments with mercury oxide must be strictly observed.
Decontamination of mercury waste
The entire filtrate and rinsed water is collected in a large cup, if necessary the solution is brought to an alkali and sodium sulfide is added to it. Black mercury sulfide HgS forms, which can be poured down the drain.

Soluble mercury salts must not be poured down the sink. The obtained mercury oxide is stored in tightly sealed jars.
Reaction of the breakdown of mercury oxide
Obtaining oxygen in the laboratory is based on the breakdown of loose compounds which contain oxygen in their composition. These substances include Berthollet’s salt, potassium permanganate, sodium hydroxide and mercury oxide. When these substances are heated, they break down with the release of oxygen. The reaction of the breakdown of mercury oxide can be demonstrated in an experiment.
For the experiment, we use a test tube of high-melting glass with a length of 17 cm and diameter of 1.5 cm, with a bent lower end of a length of 3 cm. In the lower end, place 3-5 g of red mercury oxide. In the tilted test tube fastened to a stand, place a rubber stopper with a gas tube, through which the oxygen released in heating goes to the crystallizer with water.
When mercury oxide is heated to 500 °С, we observe the release of oxygen from the gas tube, and drops of metallic mercury form on the walls of the test tube. Oxygen dissolves poorly in water, so it is collected using the method of water displacement after the complete removal of oxygen from the test tube.
After the experiment is completed, we must first take the tube out of the crystallizer with water, then turn off the burner, and only open the test tube after it has cooled down completely (mercury fumes are very poisonous). Instead of a test tube, you can use a retort with a receptacle for mercury. From 10 g of red mercury oxide, 500 ml of oxygen is obtained. The equation of the reaction of the breakdown of mercury oxide:
2HgO = 2Hg + O₂ - 2x25 kcal
Warning! Substances of this experiment are toxic and highly dangerous for your health. Do not try this at home. Only under professional supervision.