The silver mirror reaction
The scientific description of an amazing experiment
The experiment that has a beautiful effect of a mirror surface forming on glass looks very impressive. Carrying out of this reaction requires experience and patience. In our article, you can read about the necessary reagents, how to prepare the equipment, and to find out the chemical equations of reactions.
The essence of the silver mirror reaction is the formation of metallic silver layer as a result of an oxidation-reduction reaction, by the interaction of an ammonia solution of silver oxide in the presence of aldehydes.

To make a durable silver layer, you will need:
- 100ml glass flask;
- 2,5-4% ammonia solution;
- silver nitrate 2%;
- formaldehyde solution (40%).
Instead of the ammonia and silver nitrate solutions, you can also use Tollens’ reagent – an ammonia solution of silver oxide. Add 1 gram of silver nitrate to 10 drops of water (if the liquid is supposed to be stored for a long time, keep it in a dark place or in a dark glass bottle). Before the experiment, mix the solution (around 3 ml) in the proportion of 1/1 with a 10% sodium hydroxide solution. There may be a precipitate of silver, which you can dilute by slowly addition of ammonia solution. The reaction occurs at room temperature. For the successful result, the walls of the glass flask must be perfectly clean and smooth. If there are tiny particles of dirt on the walls, the obtained precipitate will appear as a crumbly layer of a black or dark grey color.
To clean the flask, use different types of alkaline solutions. First, use a caustic soda solution, and then rinse the flask with distilled water. Rinse the flask with the cleanser many times.
Why is it so important for the vessel to be clean
The colloidal particles of silver formed at the end of the experiment must firmly interlock with the glass surface, and it should not have any fat or mechanical particles on it. The distilled water does not contain salts and is ideal for the final flask rinsing. You can make it at home, but it is easier to buy it.
Equation which describes the first stage of the silver mirror reaction:
Ag₂O + 4 NH₃·Н₂О ⇄ 2[Ag(NН₃)₂]ОН + 3Н₂О
where [Ag(NН₃)₂]ОН is silver diamine hydroxide, obtained by the dissolution of the metal oxide in an ammonia solution.
NB Observe the requirements for the ammonia concentration, the reaction is possible at low concentrations!
You can examine the final stage of the reaction in the following equation:
R-CH=O + 2[Ag(NH₃)₂]OH → 2Ag↓ + R-COONH₄ + 3NH₃ + H₂O (reaction of an aldehyde R-CH=O and silver diamine hydroxide solution produce the silver sediment, an carbonic acid amide, and ammonia solution; wherein, R is an organic functional group)
It is better to carry out the second stage of the reaction by carefully heating the flask on a burner, then the process should proceed without any problems.
What can the silver mirror reaction show
This interesting chemical reaction not only allows us to examine certain states of a substance, but can also give an exact definition of aldehydes. This reaction answers the question: is there an aldehyde group in the solution or not?
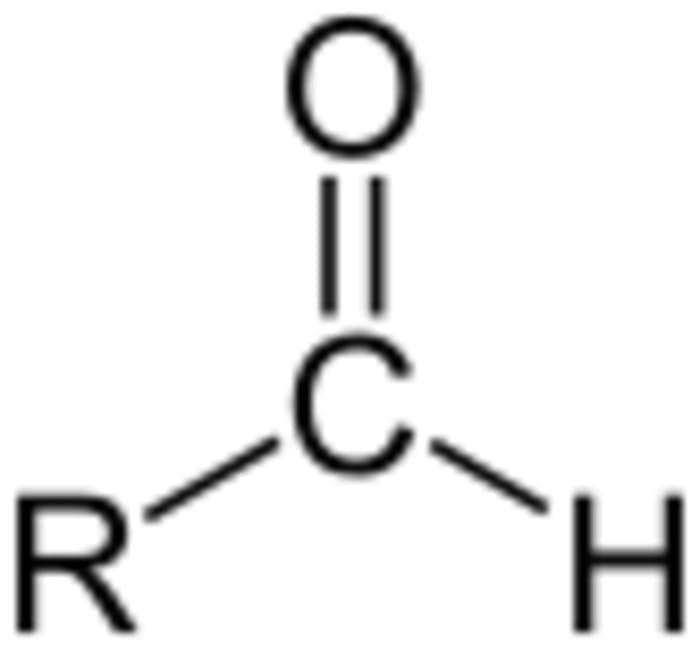
For example, due to this process, we can find out what component the solution contains: glucose or fructose. Glucose gives a positive result, a “silver mirror” is formed, while fructose contains a ketone group and cannot form a silver precipitate. To make an analysis, you should add a 10% glucose solution instead of a formaldehyde one. Let’s have a look at the equation to see why and how the dissolved silver turns into a solid precipitate:
2[Ag(NH₃)₂]OH + 3H₂O + C₆H₁₂O₆ (glucose) = 2Ag↓+ 4NH₃∙H₂O + C₆H₁₂O₇ (gluconic acid forms).