Instant crystals
Make a liquid crystallize with a light touch!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray.
- Observe safety precautions when working with boiling water.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
Take the test tube out of the beaker, open it, and add 1 mL of water. Then continue heating. Now the crystals should melt.
No problem! Just continue heating the test tube for another 5–10 minutes or until everything has melted.
This is normal – the test tube can warp slightly at high temperatures. Keep going; this won’t affect the experiment!
If there are just a few crystals, you can continue the experiment – take the test tube out of the flask and immerse a stick in the liquid. If the test tube is almost completely overgrown with crystals, repeat the experiment from step 4.
The liquid may not have cooled down completely. Wait 10 minutes and try touching the solution with the stick again. If nothing happens, repeat the experiment from the beginning.
Yes; the acetate in the same test tube can be melted and cooled multiple times.
Step-by-step instructions
Prepare a stick with sodium acetate CH3COONa · 3H2O crystals adhered to it. You’ll need it at the very end of the experiment.
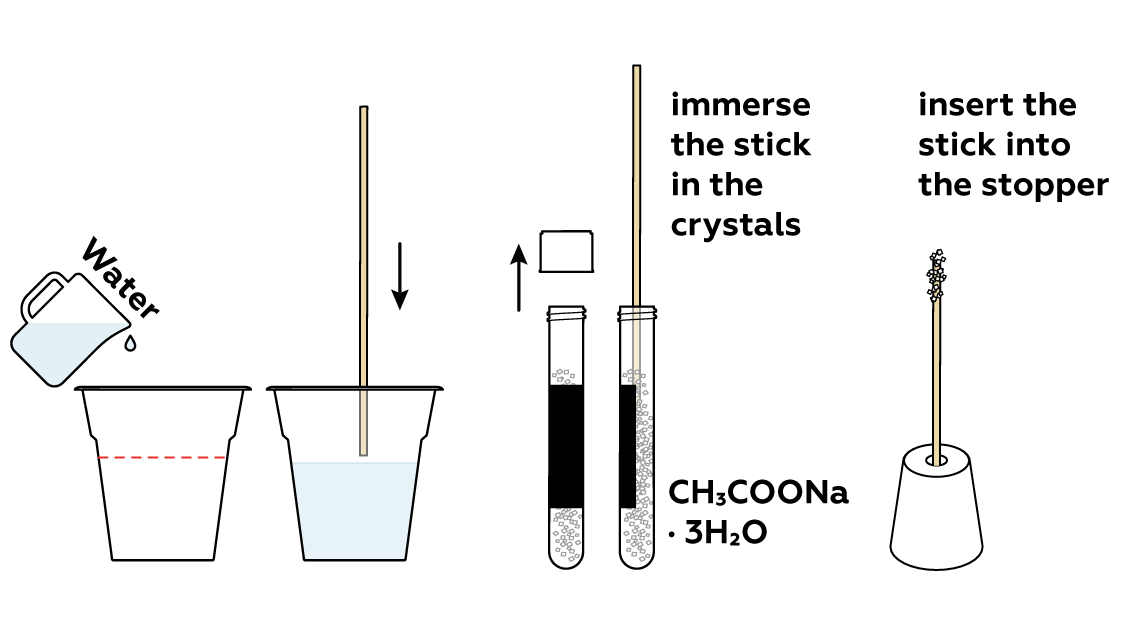
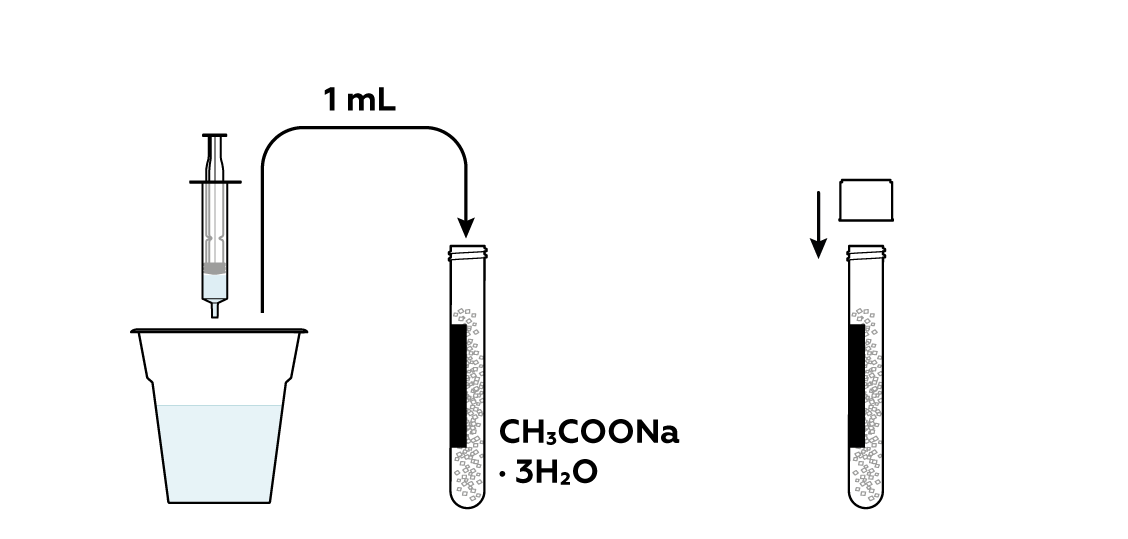
Add some water to the rest of the sodium acetate CH3COONa · 3H2O crystals. This will help them to melt.
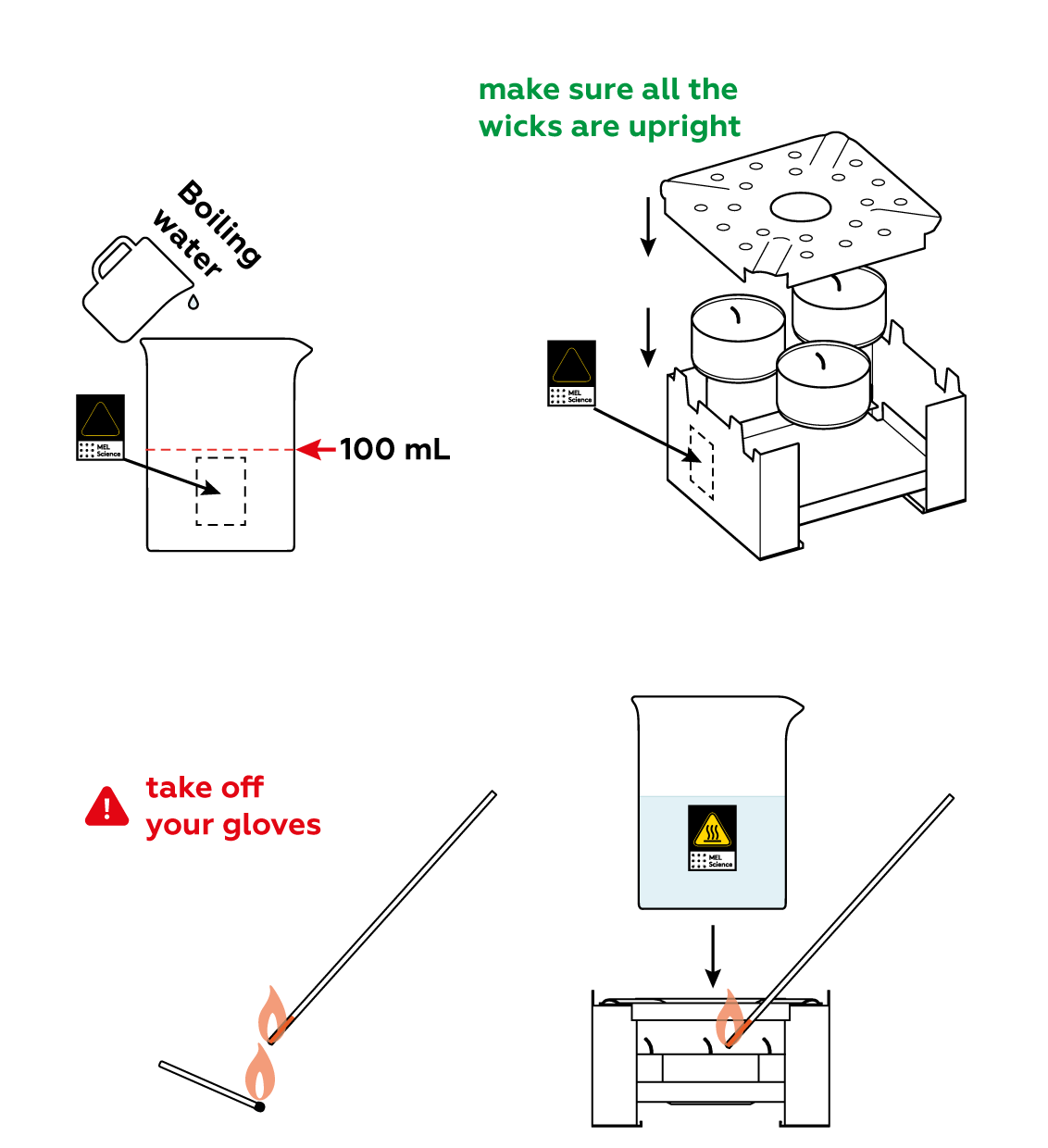
Now, build a heated bath to melt the crystals.
Place the test tube in the glass beaker and watch what happens! The crystals melt as they absorb the heat you’re providing.
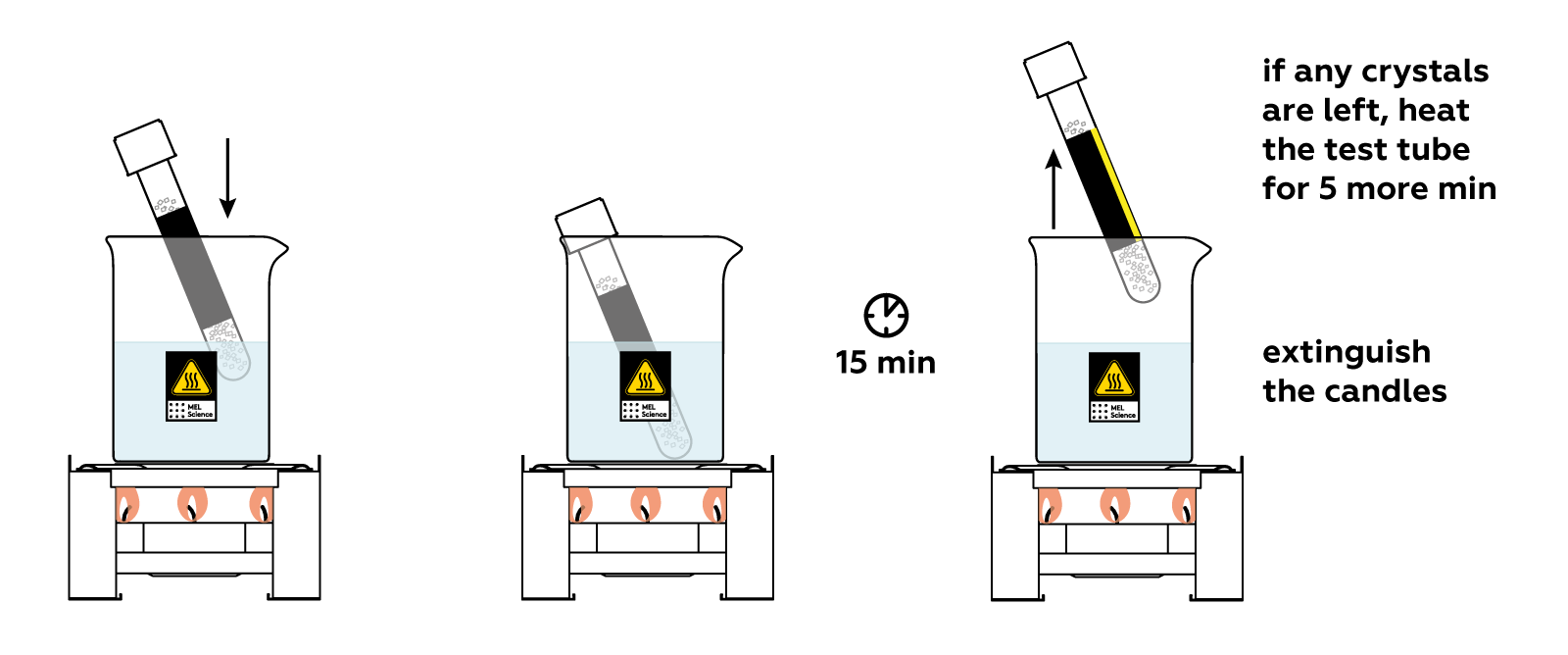
Cool the test tube quickly and carefully. At first, nothing seems to be happening. But just a soft touch of the stick triggers rapid crystal formation! And the test tube heats up dramatically.
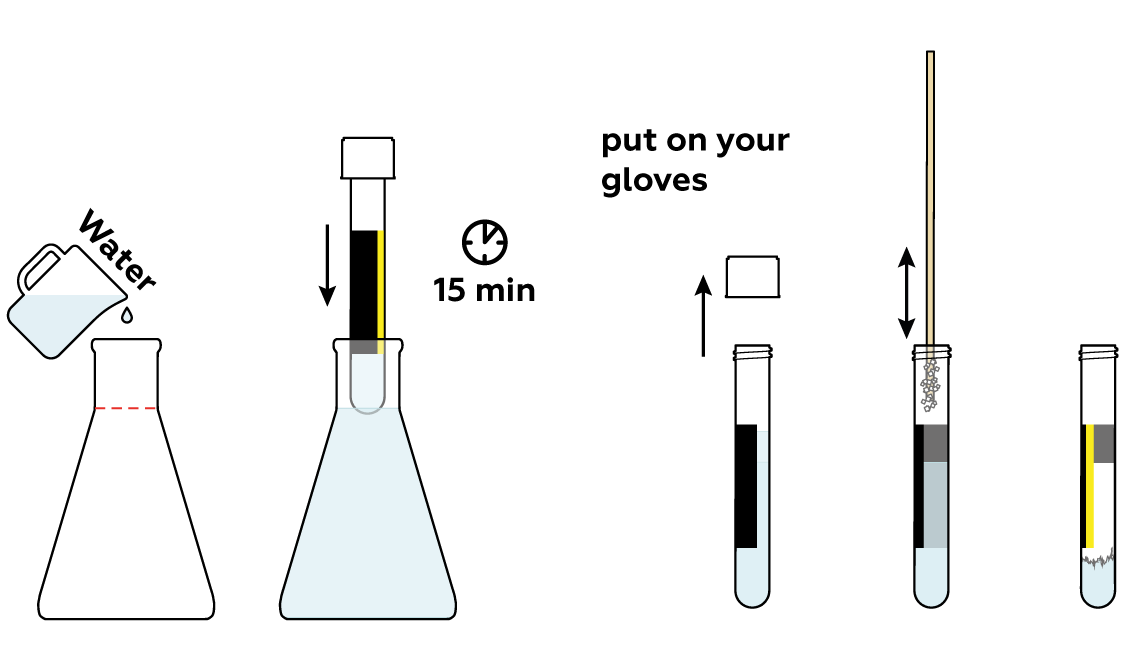
You can conduct this experiment several times! Just repeat steps 3–5.
Disposal
- Please refer to local regulations when disposing of chemicals.
- Dispose of other solid waste with household garbage.
- Pour leftover solutions down the sink. Wash with an excess of water.
Scientific description

constituting any substance aren't just sitting still—they’re constantly vibrating and bouncing around. The intensity of this movement is what we call "temperature," i.e. the faster the particles are moving
, the hotter the substance is
.
In crystals 
occupy fixed positions


. When you heat the sodium acetate crystals


and abandon their fixed "seats," eventually so much so that the crystals


When you cool the resulting liquid 



When a liquid is cool enough to crystallize but remains a liquid nonetheless, we call it a "supercooled liquid." This state is usually quite unstable, and not only actual crystals, but even a speck of dust can "remind" such a liquid to turn into a crystal.
Can water be supercooled too?
Of course! You can even try to do it at home. Check it out!
For this purpose, you will need to put a half-liter (one pint) bottle of purified water in the freezer for about three hours. When you take it out of the freezer, be gentle – the water is ready to turn into ice! Hit the bottle against the table or shake it and see how quickly it freezes!
Have you already tried it? Watch this video to see what other cool tricks you can do with supercooled water!
If supercooled liquids exist, can superheated ones exist too?
Any substance can exist in one of three states: solid, liquid, or gaseous. For water, these are ice, water, and vapor. As a rule, at normal pressure, the transition from one state to another occurs at a certain temperature: water turns into ice at 0 °C (32 °F), and becomes vapor at 100 °C (212 °F).
However, as you already know, there is an exception to this rule. When gently cooled below their freezing point, the particles that make up a liquid slow down but do not settle into their solid form. But can a liquid be just as gently heated above its boiling point and not start to evaporate? Turns out, it’s possible! And such a liquid is indeed said to be superheated.
In liquids, particles move chaotically and relatively quickly, while still remaining quite close to each other. When heated to their boiling point, they begin to move even faster and detach from the surface of the liquid, turning into a gas. However, if the liquid is heated very carefully, the particles in it will move as fast as in a gas, but will not “dare” to leave the surface. When such a liquid is disturbed, ripples form on its surface, upon which the particles instantly “realize” that it is high time for them to scatter. This phenomenon sometimes occurs when water or milk is heated in a smooth, clean cup in the microwave for a long time. The liquid can seem tranquil at first, but once something like a spoon or a tea bag is immersed in it, the disturbance can trigger the liquid to boil vigorously or even explode! If hot droplets land on the skin, they can cause serious burns, so be careful when heating liquids in the microwave for extended periods of time!
That’s interesting!
Supercooled water in nature
The supercooling of water underlies the natural phenomenon of freezing rain. If it rains at temperatures below 0 °C (32 °F), the raindrops are supercooled. When they come into contact with any surfaces, such as the ground, tree branches, or power lines, they instantly freeze, forming a thin film of ice. This can cause a plethora of problems: power lines and tree branches can break under the weight of the ice, and ice on the roads can lead to car accidents.

