Anthocyanins
Explore natural pH indicators!
Reagents
Safety
- Put on protective eyewear. Put protective gloves on before step 2.
- Conduct the experiment on the plastic tray.
- Observe safety precautions when working with boiling water.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
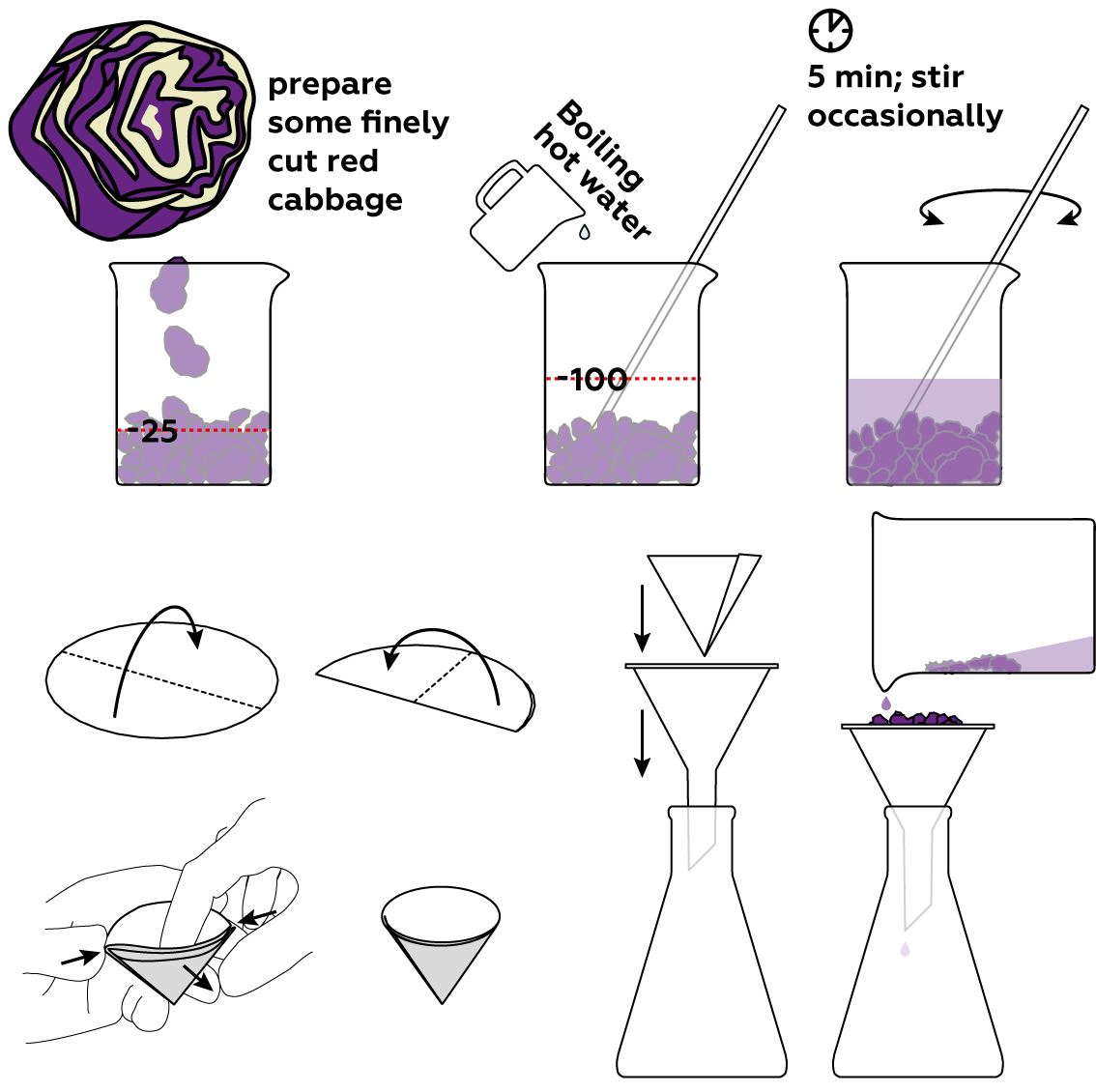
We recommend using a knife to cut the cabbage into small pieces. The smaller the pieces, the greater their contact area with water, and the better your results will be!
This can happen if you use cold or warm water. You need to use boiling water for this experiment! It’s probably best to start the experiment over.
Pour everything in the flask back into the beaker. Take a new sheet of filter paper and fold it according to the instructions. Filter the cabbage out a second time.
You can repeat this experiment with cherries, black grapes, blackberries, and red rose petals. Note that if you use red rose petals, you need to fill the beaker with them above the 25 mL mark because the petals are quite voluminous. If you opt for black grapes, cherries, or blackberries, you may have more difficulty or spend significantly more time filtering the solution. Ultimately, though, you should still get the desired results!
The color of the resulting solution in the beaker and in the vials depends on what object (flower, berry, or vegetable) you chose for the experiment. Moreover, the composition of the substances you extract depends somewhat on the individual plant they came from, and even the time of year the plant was harvested! Therefore, the colors will never be exactly identical to what is shown in the instructions.
Step-by-step instructions
Red cabbage owes its color to compounds known as anthocyanins. Anthocyanins are soluble in water, so you can extract them easily. You can also use crushed blackberries, red rose petals, or red grape skin.
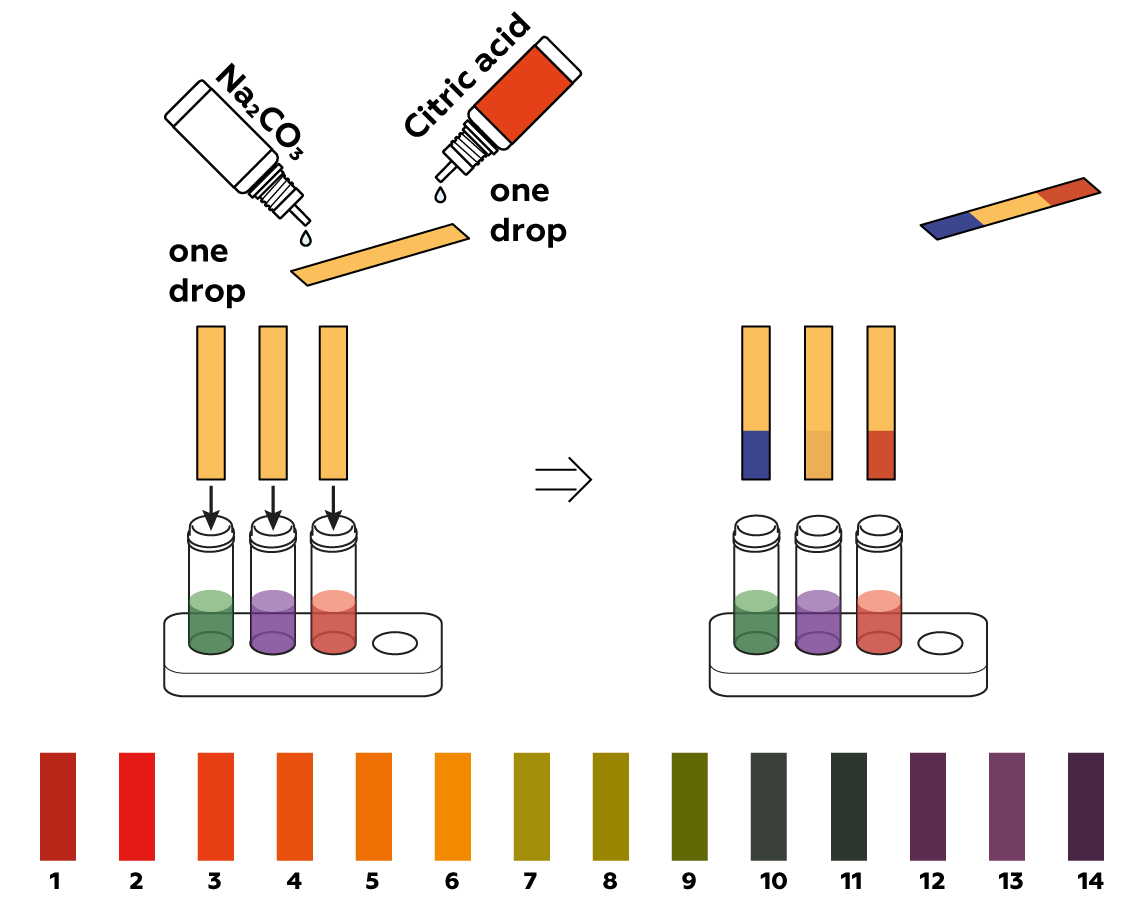
Citric acid increases the acidity of solutions, while sodium carbonate Na2CO3 makes solutions more basic. Anthocyanins can change colors dramatically depending on the acidity of the medium they’re in.

Chemists use an acidity unit called pH. It is often measured using universal pH indicator strips, which change colors like anthocyanins. Compared to these strips, anthocyanins are much less consistent—anthocyanins from different sources can display different colors at the same pH level. Test the accuracy of your cabbage juice using commercial pH indicator strips!
Disposal
Dispose of solid waste together with household garbage. Pour solutions down the sink and wash with an excess of water.
Scientific description
What is shown in the photograph that is found with the experiment instructions?
It is a macro photograph illustrating how a petal changes its color when it is exposed to sodium carbonate (on the left). The picture shows individual plant cells – each of them has a different concentration of hydrogen ions H+ and, therefore, presents a different color. In this experiment, we can create all the colors shown in the picture – we’ll just need several dark flower petals (such as - rose, petunia, pansy), some warm water, and solutions of sodium hydrogen sulfate NaHSO4 and sodium carbonate Na2CO3!
What are anthocyanins?
Anthocyanins are a whole family of plant pigments. They are present in lilac, red, purple, violet or even black flower petals. Anthocyanins are also found in fruits and vegetables, as well as some leaves. Cold weather causes these purple pigments to absorb sunlight more intensely, which, in turn, raises the core temperature of the plant compared to that of the ambient air. This protects the plant from cold temperatures. In hot weather or at high altitudes, anthocyanins protect the plant cells by absorbing excessive ultraviolet radiation. Furthermore, a vivid petal coloration makes it easier for insects to find the flowers and pollinate them.
Why does adding sodium hydrogen sulfate NaHSO4 make the solution red?
Adding NaHSO4 to water increases the number of protons H+ in the solution. In other words, we increase the acidity of the medium because sodium hydrogen sulfate dissociates in water, or, in other words, it breaks down into individual ions:
NaHSO4 → HSO4- + Na+
HSO4- ↔ SO42- + H+
In turn, the H+ protons react with the anthocyanin molecules transforming them from the neutral into cationic form. The cationic form of anthocyanins has a bright red color.
Why does the solution change its color?
The color of anthocyanins is determined by the concentration of hydrogen ions H+. When we add the sodium carbonate Na2CO3 solution, the H+ concentration drops. A decrease in the number of H+ causes a pigment color change, first to purple and then to blue and dark green.
Why does the basic solution turn yellow with time?
Anthocyanins are unstable in a basic environment, and so they gradually decompose. The decomposition process produces yellow-colored substances called chalcones. This process is quite slow, allowing us to track how a solution changes its color from blue to various shades of green and finally to yellow.
What flowers have suitable petals for this experiment?
The best petals would be brightly colored dark petals of red, purple, blue, or violet. You are particularly lucky if you can get your hands on almost black petals from either petunia, roses, irises, African violets, tulips, or lilies. These flowers contain a maximum concentration of anthocyanins.
Interestingly, flowers are not the only substrate you can use in this experiment. It will work with vegetable juices such as beet, red onion or red cabbage. The best would be red cabbage juice as it shows the richest color palette in this experiment. Other suitable sources for anthocyanins are berries – blackcurrant or blueberry –as well as red grapes.
That’s interesting!
The Violet Accident
British scientist Robert Boyle (1627–1691) made a number of remarkable discoveries in chemistry. Interestingly, one of these discoveries involved the beautiful flowers known as violets.
One day, Boyle brought a bouquet of violets to his laboratory. His assistant, who was performing an experiment at the time, accidentally splashed some hydrochloric acid on the flowers. Worried that the acid would harm the plants, the assistant moved to rinse them with water, but Boyle suddenly stopped him.
The scientist’s attention was fixed on the violets. The places where acid had splashed the petals had turned from purple to red. Boyle was intrigued. “Would alkalis affect the petals, too?” he wondered and applied some alkali to a flower. This time the petals turned green! Experimenting with different plants, Boyle observed that some of them changed colors when exposed to acids and alkalis. He called these plants indicators.
By the way, the violet color of the petals is produced by anthocyanins – pigments that absorb all light waves except violet. These vibrant pigments help attract bees, butterflies, and other pollinators, facilitating the flower’s reproduction.