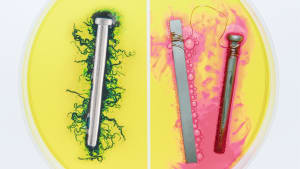
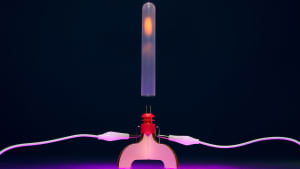
Mg – magnesium – is a light, shiny metal with a silvery-white color. Pure magnesium is soft and fragile, but its alloys with aluminum, zinc, copper, and silicon are lightweight and strong. You can therefore find housings made of magnesium alloys in such devices as mobile phones, video cameras, and laptops. It is also used in the construction of automobile parts, aircraft parts, and even missiles. Magnesium can be found in many medicines. It is used as an anticonvulsant, laxative, sedative, cardiac, and antispasmodic. It is also used to regulate the acidity of gastric juices and to treat injuries and joints.
Similar reagents
Experiments
Experiments with similar reagents
Hazard information
Hazard statements
- In contact with water releases flammable gas.
- Flammable solid.
- Self-heating in large quantities; may catch fire.
Precautionary statements
- Store in a dry place. Store in a closed container.
- Keep container tightly closed.
- Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking.
- In case of fire: Use dry sand, dry chemical or alcohol-resistant foam to extinguish.