Copper complexes
Discover the bright colors of copper complexes!
Reagents
Safety
- Put on protective gloves and eyewear.
- Conduct the experiment on the plastic tray and in a well-ventilated area.
- Keep vials away from nose and mouth.
- Avoid inhaling ammonia gas from the vial with ammonium carbonate.
- Do not allow chemicals to come into contact with the eyes or mouth.
- Keep young children, animals and those not wearing eye protection away from the experimental area.
- Store this experimental set out of reach of children under 12 years of age.
- Clean all equipment after use.
- Make sure that all containers are fully closed and properly stored after use.
- Ensure that all empty containers are disposed of properly.
- Do not use any equipment which has not been supplied with the set or recommended in the instructions for use.
- Do not replace foodstuffs in original container. Dispose of immediately.
- In case of eye contact: Wash out eye with plenty of water, holding eye open if necessary. Seek immediate medical advice.
- If swallowed: Wash out mouth with water, drink some fresh water. Do not induce vomiting. Seek immediate medical advice.
- In case of inhalation: Remove person to fresh air.
- In case of skin contact and burns: Wash affected area with plenty of water for at least 10 minutes.
- In case of doubt, seek medical advice without delay. Take the chemical and its container with you.
- In case of injury always seek medical advice.
- The incorrect use of chemicals can cause injury and damage to health. Only carry out those experiments which are listed in the instructions.
- This experimental set is for use only by children over 12 years.
- Because children’s abilities vary so much, even within age groups, supervising adults should exercise discretion as to which experiments are suitable and safe for them. The instructions should enable supervisors to assess any experiment to establish its suitability for a particular child.
- The supervising adult should discuss the warnings and safety information with the child or children before commencing the experiments. Particular attention should be paid to the safe handling of acids, alkalis and flammable liquids.
- The area surrounding the experiment should be kept clear of any obstructions and away from the storage of food. It should be well lit and ventilated and close to a water supply. A solid table with a heat resistant top should be provided
- Substances in non-reclosable packaging should be used up (completely) during the course of one experiment, i.e. after opening the package.
FAQ and troubleshooting
Don't worry! Continue the experiment and see what happens. When you’re done, you can repeat the experiment using the correct measurements and compare your results.
Yes, it is! This reaction releases carbon dioxide gas CO2, which creates the bubbles you see. Moreover, a small amount of ammonia NH3 is released too, so you might sense a slight odor.
It’s possible that you mixed up the reagents while doing the experiment. Repeat the experiment, keeping careful track of which reagent you use.
To dispose of the solutions, drain them in the sink and rinse them down with plenty of water. You might sense a slight odor of NH3 and SO2. If you do, air the room out after the experiment.
Step-by-step instructions
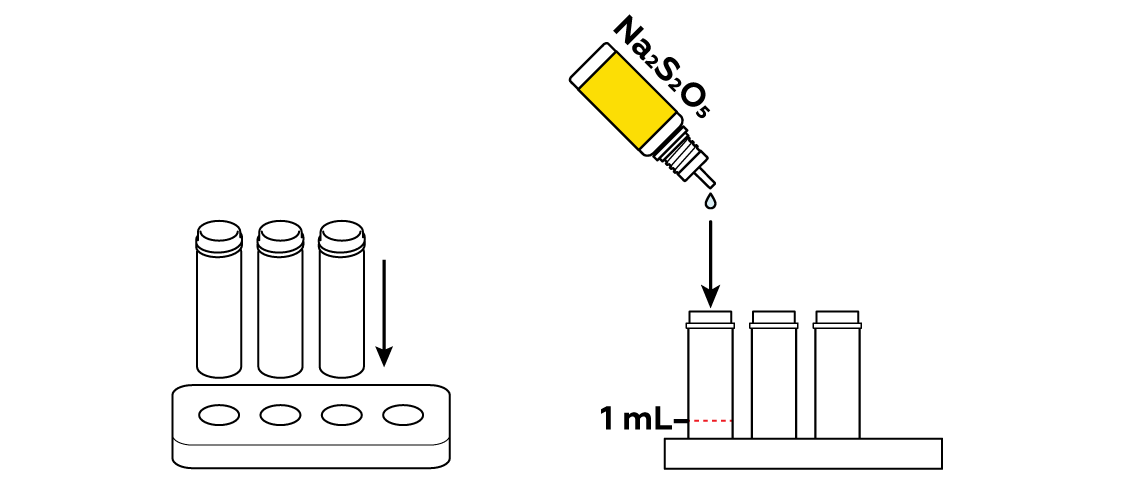
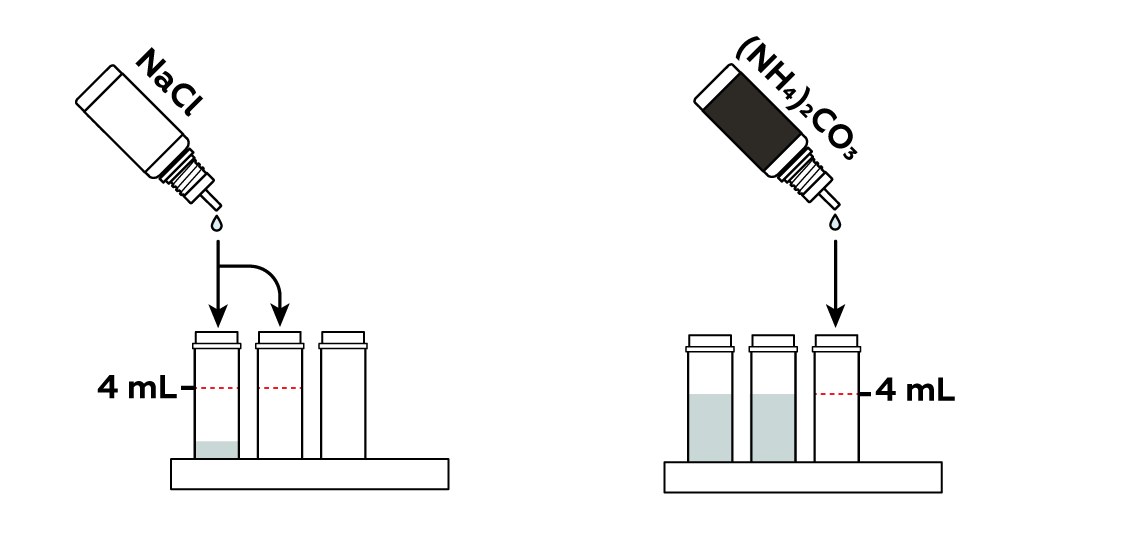
Pour three colorless compounds into separate vials.
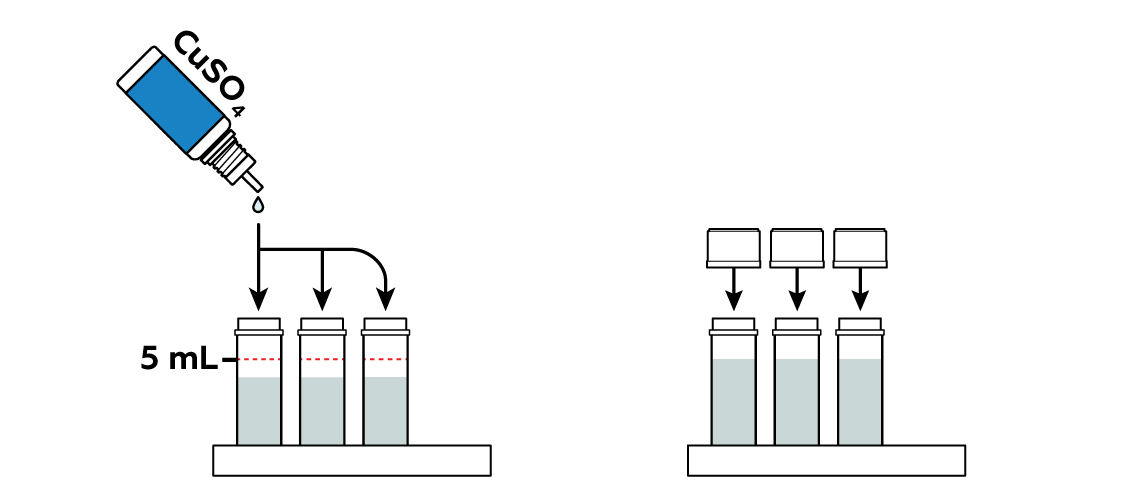
Add copper sulfate CuSO4 to the vials.
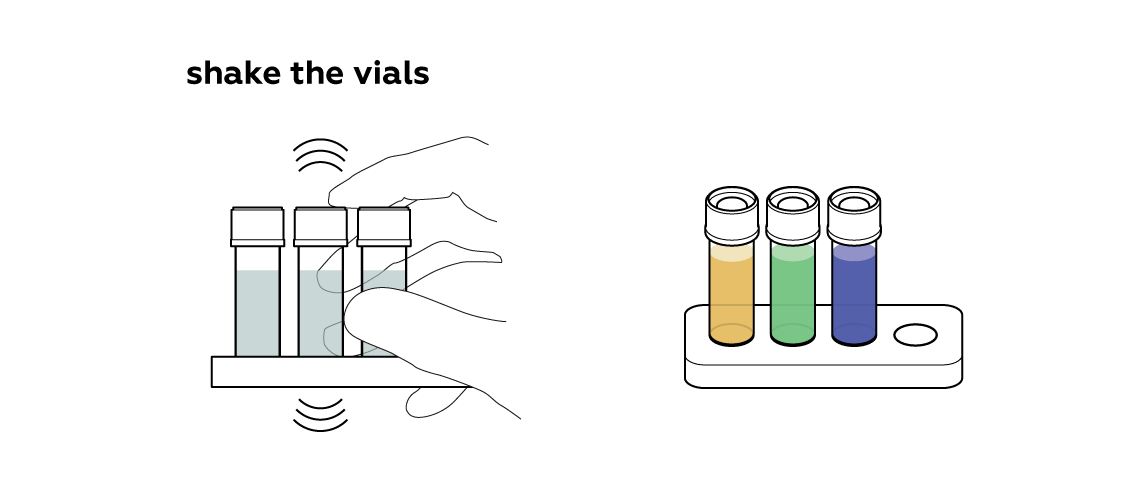
Mix the solutions.
Disposal
Dispose of solid waste together with household garbage. Pour solutions down the sink and wash with an excess of water.
Scientific description
The copper sulfate CuSO4 solution has a nice blue color, but this color changes when you mix it with other compounds. Why is that? As it turns out, the color mostly comes from the interactions of copper particles—copper ions Cu2+ with their surroundings. In the CuSO4 solution, for example, they are surrounded by H2O molecules
, and this arrangement results in this vibrant blue. By the way, copper sulfate crystals owe their blue color to water as well, as you can see in another experiment from this set.
Ammonium carbonate (NH4)2CO3 yields some ammonia NH3 molecules
in the solution, which easily replace the water molecules around Cu2+
. This arrangement, known as a copper-ammonia complex
, produces a rich dark-blue color.
The sodium chloride NaCl solution contains chloride ions Cl\-
. When Cu2+
is surrounded
by these, you can observe a really beautiful green.
What disodium disulfite Na2S2O5 does is a bit trickier. On the one hand, it reacts with Cu2+
, turning them into Cu1+
. On the other hand, it easily yields SO2 molecules
. The latter can form a complex
with Cu+
, and it gives the solution a yellow-orange tint.